
Environmental and occupational determinants of lung cancer
Background and rationale
Lung cancer is a leading cause of morbidity and mortality globally, accounting for 2,094 million cases and 1.8 million deaths per year (1). Lung cancer has become the most common cancer in the world for the last several decades and showing an increase in incidence in the developing countries (2). Lung cancer, unfortunately does not become clinically apparent until it reaches an advanced stage and more than 75% of lung cancers are diagnosed when disease is advanced or metastatic (3). In India, approximately 114,000 new lung cancer cases are reported each year (4).
According to GLOBOCAN 2018, lung cancer represented 11.6% of all types of cancers, out of which 80% were attributed to smoking (5-7). Outdoor air pollution caused 108,000 lung cancer deaths (8); solid fuels, used more in developing countries for cooking and heating, caused 36,000 lung cancer deaths (8,9); second-hand smoke was estimated to cause 21,000 lung cancer deaths (10).
The chances of getting lung cancer has increased with increase in household air pollution (11,12). Only 1 out of 10 patients diagnosed with lung cancer will survive the following 5 years (13). It has been noticed that smoking prevalence has declined in many developed countries (14) however; it is increasing in developing countries and also among women (15). In developing countries lung cancer is usually detected at an advanced stage (16,17) and this puts a greater economic impact on middle- or low-income families. Lung cancer has multiple risk factors, which are combination of the genetic and external factors (environmental & occupational) and the proportion of lung cancer cases attributable to preventable risk factors vary greatly across countries (18,19). The resulting evidence from various research studies, causally associate lung cancer with active and passive smoking, a variety of occupational agents, and indoor and outdoor air pollution (20).
Lung cancer can be preventable, because most risk factors of lung cancer can be prevented if people have sufficient knowledge about that and the government should make specific policy and guideline on these environmental and occupational risk factors which attribute to lung cancer such as tobacco (smoke and smokeless) consumption (21). Therefore, environmental and occupational interventions are a solution for diminishing the incidence and mortality of lung cancer, by abolishing these risk factors by means of primary prevention.
Environmental factors can be physical (ionizing and non-ionizing radiations such as exposure to radon or ultraviolet (UV) radiation, respectively), chemical (such as asbestos, dioxins, metals (such as arsenic chromium, nickel, cobalt, etc.) and other pollutants found in industrial emissions, house hold smoke and second-hand smoke. Parents who have occupational exposures may increase the risk of cancer in their progeny (22). Lung cancer is strongly linked to environmental & occupational exposure (23).
Exposure may be widespread, starting from outdoor and indoor air pollution, or could be restricted to an area close to a certain industrial site. These exposures have been associated with a variety of neoplasms, but most important among these is lung cancer. Occupational health risks are also directly related to physical, chemical and biological factors that present in the environment. Lung cancer has most commonly been linked with occupational exposures and for which evidence is strong (24). Exposure limit of various environmental and occupational risk factors are summarized in Table 1.
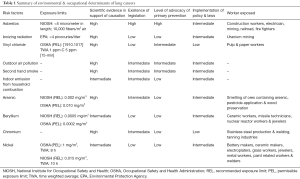
Full table
A WHO study found that at least 1.7 million cancer deaths annually could be prevented through healthy working and living environments (25). Changes in individual behavior are facilitated by wider contextual changes in the environments where individuals live and work. Respiratory diseases, for instance, are tied to lung cancer risk factors such as smoking and air pollution. Therefore, primary prevention can achieve a number of primary end points (21).
Environmental determinants of lung cancer
Chemical exposures
There is a strong evidence that exposure to industrial and manufacturing chemicals and harmful pesticides or aflatoxin can compose of cancer-trigger factors that present in our living and working environments. Agricultural and public health workers are mainly exposed to these hazardous pesticides during handling, dilution and application. Exposure is mainly by skin and by the respirable routes during application. Occupational pesticide usage is related to lung cancer in some cases but not all. In a study by Bonner et al., the occupational hazard ratio was elevated in the highest exposure category of lifetime to 3 chemicals used as pesticides, associating it with lung cancer incidence (26).
In a follow-up to this study, Alavanja et al. evaluated the use of 43 pesticides and 654 lung cancer cases after 10 years of additional follow-up in the Agricultural Health Study (AHS), a prospective cohort study comprising 57,310 pesticide applicators from Iowa and North Carolina. The authors found possible new associations for chlorimuron ethyl and parathion with lung cancer that have not been previously observed in the AHS, and are of the view that it needs to be further studied. Above cited study and several other studies along with this, support the theory that pesticides are indeed a major determinant of lung cancer (27).
Chlorophenols (CPs), dioxin compounds and related phenoxyacetic acids (PAs) are pesticide groups that have carcinogenetic effect on lung (28-30). Various studies reported a wide range of lung cancer in workplace site (31).
Meta-analysis with five cohort studies (32-36) with six reports, investigated the causes of lung cancer among workers of CPs related compounds (CPsR) plants. Collins et al. (37) evaluated two individual investigations with different types of CPs exposure. Among the studies, one multicenter analysis from IARC publications included 36 studies from previous reports (32). Overall, a total of 27,865 workers in CPsR production were involved in the meta-analysis. Five papers with six reports were included in the final analysis. The standardized mortality rate (SMR) for lung cancer from the random model was 1.18 [95% confidence interval (CI): 1.03–1.35, P=0.014] (38)
Many research studies observed no statistically significant odds ratio (OR) by duration of pesticide exposure, intensity of pesticide exposure, and cumulative pesticide exposures with lung cancer mortality in both smokers and nonsmokers (24,27,39).
Outdoor air pollution
Many air pollutants are released into the air from mining and other industries, and from municipal waste sites and inadequate domestic incineration (40). Also, motor vehicles can add notably to air pollution in urban areas. Some substances that are present in vehicular emission exhaust are classified as carcinogenic to humans (group 1) and probably carcinogenic to humans (group 2A) (41). Various research studies have found a higher lung cancer risk among urban people in comparison to persons living in rural areas (42). In U.S., Europe, Russia and East Asia, agricultural emissions make the largest relative contribution to particulate matter (PM)2.5. Emission scenario indicates that the contribution of outdoor air pollution to premature mortality could double by 2050 (43). The risk of developing lung cancer increases as the level of PM2.5 in the air increases.
Indoor air pollution from household combustion
Burning coal inside home for the purpose of heating or cooking produces PM and gas emissions that may contain a number of cancer trigger factors, such as benzene, carbon monoxide, formaldehyde, and polycyclic aromatic hydrocarbons (PAHs). Indoor air pollution from household combustion of coal is carcinogenic to humans (group 1) (44). There is rich evidence that proved carcinogenicity in humans from household combustion of coal (12,45). Increasing level of smoke inside the home associated with an increasing risk of lung cancer (P<0.05) (OR 1.62; 95% CI: 1.14–2.32) (46). Another research study findings showed that Individuals who carried the T-genotype of HIF-1α rs2057482 were more susceptible to small cell carcinoma (OR 1.725; 95% CI: 1.047–2.842) (47).
Second-hand smoke
Tobacco and second-hand smoke are known to be a human carcinogen (48). Tobacco smoking is attributed to lung cancer in humans. The risk of death from lung cancer increases with increasing duration of smoking and with increasing numbers of cigarettes smoked. Studies support an association of environmental (passive or secondhand) tobacco smoke with cancer of the lung (49,50).
Many epidemiological studies, including case-control studies, have demonstrated increased risk for developing lung cancer following prolonged exposure to environmental tobacco smoke either second-hand aerosol from tobacco or electronic cigarettes (51). Exposure to environmental tobacco smoke during childhood is strongly associated with lung cancer (OR 3.9; 95% CI: 1.9–8.2) according to one research study (52). According to another study, those who had never smoked, both among men (P=0.02) and women (P=0.001), presented more frequently with adenocarcinoma histology (53).
Asbestos
Asbestos are naturally occurring fibrous silicates with important commercial use in acoustical and thermal insulation. They can be divided into two groups: chrysotile and the group of amphiboles, including amosite, crocidolite, anthophyllite, actinolite and tremolite fibres. All types of asbestos are carcinogenic and can cause lung cancer and mesothelioma. Biological effects of amphiboles on the pleura and peritoneum seem to be stronger than those of chrysotile. There are many epidemiological studies available on asbestos-exposed workers, but only few on the health effects of household and residential exposure to asbestos. The main household exposure concerns are to the immediate family members of asbestos workers and arise from dust brought from workplace on clothes, while household sources of asbestos exposure are represented by the degradation, installation, removal and repair of asbestos-containing products. Residential exposure mainly results from asbestos mining related outdoor pollution or manufacturing in the nearby places in addition to natural exposure from the erosion of asbestos or asbestiform rocks. Assessing the non-occupational exposure to asbestos is difficult, since levels are generally low, and the duration and frequency of exposure and the type of exposure is seldom well defined. According to IARC, all forms of asbestos (chrysotile, crocidolite, amosite, tremolite, actinolite and anthophyllite) are carcinogenic to humans (group 1). Environmental Protection Agency (EPA) also classified asbestos as Group A carcinogen to human (54).
A large number of epidemiological and occupational studies have reported that exposure to asbestos via inhalation causes lung cancer and mesothelioma (55-59). Study on workers with heavy exposure to asbestos showed an increased risk of lung cancer [relative risk (RR) 1.74; 95% CI: 1.25–2.41] and a similar increase in risk to those with low exposure up to 20 years (RR 0.94; 95% CI: 0.77–1.15) (60). Carcinogenicity of asbestos depends upon its length of fiber. Long and intermediate length-range asbestos fibers (>5 µm) are proven to be more carcinogenic to humans than short fibers. An autopsy study of Italian shipyard workers showed that chrysotile was the most common type of asbestos fiber in asbestos-related lung plaques. Most of those fibers were found to be of 8 mm in length (61). Use of asbestos has continued in much of Asia, Africa and India has been a major consumer of asbestos after China (62).
Metals
Arsenic is both an environmental and occupational lung carcinogen which most commonly exists in the form of arsenite and arsenate. Occupational exposures occur primarily among workers who work and inhale dust from lead, gold, and copper ore mines and smelters. The IARC concluded that arsenic exposure via inhalation increases lung cancer risk (63). Many case-control studies were performed in Argentina, Bangladesh, Chile, and Taiwan, that examined the associations between higher concentrations (e.g., >100 µg/L) of arsenic in drinking water and lung cancer. It is not known if the lack of evidence of an association below 100 µg/L is the result of a threshold effect or not (64).
According to EPA & IARC, it is probable human carcinogen and there is sufficient evidence that beryllium compound causes lung cancer (65-69). Many research studies found a significantly increased risk of lung cancer associated with hexavalent chromium (OR 1.94; 95% CI: 1.10–3.43; P=0.015) (70). Cumulative hexavalent chromium exposure was associated with an increased lung cancer risk (71). Environment protection agency has not evaluated nickel as a class of compounds for potential human carcinogenicity (72). Occupational exposure to nickel compounds could be attributed to lung cancer (73-75).
Vinyl chloride
In poly vinyl chloride packers and baggers, the risk for lung cancer increased significantly with exposure to poly vinyl chloride dust (OR 1.2003; 95% CI: 1.0772–1.3469; P=0.0010) (76), and vinyl chloride (OR 1.05; 95% CI: 0.68–1.62) (77). However, the exposure levels for the majority of the population are very low (78-80).
Ionizing radiation
Natural radiation comes from many sources including many naturally-occurring radioactive materials found in soil, water and air, like Radon, a naturally-occurring gas which comes from rock and soil, and is the main source of natural radiation. Ionizing radiation includes radon, X-rays and gamma rays, but radon represents a far smaller risk for lung cancer (81,82). Results of many research studies provided direct evidence of an association between residential radon and lung cancer risk, a finding predicted by extrapolation of results from occupational studies on radon-exposed underground miners. The excess OR (EOR) was 0.10 per Bq/m3 with 95% confidence limits −0.01 to 0.26 (83) & RR was 0.08 (95% CI: 0.03–0.16; P=0.0007) (84).
Contamination by uranium is often cited as a risk factor in ionizing radiation epidemiology, but the dose-effect relationship is rarely studied and retrospective assessment of individual exposure is generally insufficient. Moreover, it is difficult to distinguish between uranium radiotoxicity, its chemical toxicity and the radiotoxicity of its progeny (85).
Occupational determinants of lung cancer
Bartenders
In Canadian Labour Force Ten percent sample study, a total of 9,739 deaths from cancer between 1965 and 1979 were identified. Based on the criteria of strength of association, bartenders with lung cancer seem most likely to be caused by excess smoking and/or alcohol consumption (86). Secondhand smoke (SHS) exposure has been consistently identified as a public health hazard and cause of lung cancer. The U.S. EPA classified SHS as a group A carcinogen (87) and the IARC also classified SHS as carcinogenic to humans. Siegel et al. (88) demonstrated that the nicotine concentrations in the 5 B’s (bars, bowling alleys, billiard halls, betting establishments, and bingo parlours) were 2.4 to 18.5 times higher than in offices or residences, and found to be 1.5 to 11.7 times higher than in restaurants.
Ceramic industry
In a study of 30-year mortality and respiratory morbidity of refractory ceramic fiber workers, authors showed no increase in SMR for lung cancer (89). Similarly, Meijers and co-authors suggested that the disease process resulting in silicosis in the ceramic industry carries an increased risk of lung cancer, which is supportive of a nongenotoxic pathway. The authors found no increase in overall and cause-specific mortality in the total group of ceramic workers, and a statistically significant cumulative dose-response relation for silica exposure and lung cancer also did not emerge (90). Overall, inference can be made that lung cancer might be merely indirectly related with exposure to silica, although the dose may differ for silicotic and nonsilicotic individuals (91).
Coal gasification and coke production
A significant risk of lung cancer was observed among the coal/coke and related product industry with RR of 1.55 (95% CI: 1.01–2.37) (92). Excess of lung cancer in association with coal gasification was found (93,94). In addition to PAHs, workers in coal gasification may be exposed to many compounds, including asbestos, silica, arsenic, cadmium, lead, nickel, hydrocarbons, sulfur dioxide, sulfuric acid, aldehydes, etc. In UK, Helmet respirator, the Airstream helmet, was introduced with the aim of reducing exposure to PAH aerosols and consequent lung cancer risks for workers on the coke oven tops.
Construction industry
Approximately 70 different substances are listed in NIOSH to which U.S. construction workers are potentially exposed (95).
Many epidemiological studies conducted in different countries, found significantly raised mortality rate for lung cancer among construction workers (96-105) although some failed to find the association (106-109). Elevated mortality for lung cancer has also been reported for several specific construction trades such as bricklayers (97,100,103,105,109-115); craft workers (110,112); electricians (100,113); carpenters (97,99,100,103,111,115-117); painters (100,105,111,117); operating engineers (118); roofers, water proofers, allied workers (97,103,117); and insulation workers (97,100,104,109,118).
In one of the studies, it was observed that construction workers were more at risk of developing lung cancer when they were exposed for longer duration. This risk has not been seen in supervisors, engineers and higher officials working in construction as their exposure was not as high as others working at construction sites.
Glass factory
Carcinogenic effects of exposure to man-made mineral fibers (glass filaments, ceramic fibers, and slag wool) have been demonstrated. In humans, slag wool and glass production workers have been shown to have elevated lung cancer risks (119,120). Population-based case-control studies in Sweden (121) and Canada (122) also found increased risks for lung cancer in glass-workers.
Mason
Bricklayers may be exposed to several lung carcinogens, including asbestos and crystalline silica. That is why bricklayers and allied craft workers are at risk from diseases associated with heavy exposure to inorganic dust, mainly lung cancer. SMRs and proportionate mortality ratios (PMRs) for lung cancer (SMR =158; 130–190 and PMR =144) were significantly elevated in these workers (110,112). In an Italian case-controlled study, authors found increased lung cancer risk for bricklayers (OR 1.57; 95% CI: 1.12–2.21; 147 cases, 81 controls). There were increased risk for squamous cell (OR 2.03; 95% CI: 1.32–3.13, 56 exposed cases) and small cell carcinomas (OR 2.29; 95% CI: 1.29–4.07, 21 exposed cases), while not for adenocarcinoma (123) among bricklayers (OR 1.06; 95% CI: 0.68–1.65, 41 exposed cases).
Painter
A large number of workers are engaged in paint-related occupations and there is increasing concern regarding health effects from paint-related exposures. Several hazardous chemicals (including benzene, phthalates, chromium, and lead oxides) have been reduced or replaced in paint in some countries, although they are still used elsewhere. There have been indications that painters are at an excess risk for cancer, and in particular lung cancer (124,125). Raman Kumar et al, showed painters had an OR of lung cancer of 1.3 (95% CI: 0.9–2.2). Regarding exposures, ORs were: for wood varnishes and stains, 1.6 (95% CI: 1.0–2.3); for wood and gypsum paints, 1.3 (95% CI: 0.9–1.7); and for metal coatings, 1.1 (95% CI: 0.8–1.6) thereby suggesting that some exposures in paint-related occupations, particularly wood varnishes and stains, increase the risk of lung cancer (126).
Rubber industry
According to IARC, occupational exposures in the rubber manufacturing industry have been considered carcinogenic to humans (127,128). Two compounds which are largely involved in production of tyres and rubber goods are 1,3-butadiene and benzene, and both of them are established as carcinogens to humans (129,130). Several types of chemical compounds are used in production of tyres and rubber goods which can be carcinogenic to humans. In a recent meta-analysis performed on observational studies (published until April 2016) on occupational exposures in the rubber manufacturing industry and cancer risk, showed a borderline statistically significant increased risk (SRR =1.08; 95% CI: 0.99–1.17) (131).
Sandblasting
Crystalline silica exposure occurs in the workers employed in foundries, stonework, sandblasting, and potteries. Besides its well-known relation to silicosis, silica exposure in recent years has been associated with lung cancer. The IARC determined that inhaled crystalline silica from occupational sources is a definite (group 1) human carcinogen (132). Steenland et al. found a 60 percent excess of lung cancer overall, in the silica exposed cohort when compared with the U.S. population (SMR =1.60, 95% CI: 1.31–1.93). The cohort experienced high mortality for all causes combined (SMR =1.23, 95% CI: 1.16–1.31), in part due to elevations in causes related to silica exposure (133). A case report by Güngen et al. demonstrated a case of lung adenocarcinoma in 35-year-old female with history of working in denim sandblasting for 18 months (134). On molecular level, critical role of the inflammatory mediator LTB4 and its receptor BLT1 has been demonstrated in promoting the silica mediated lung tumor growth (135).
Truck drivers
Trucking industry workers who have been regularly exposed to diesel exhaust (DE) have an elevated risk of lung cancer with each increasing year of work. As per IARC, DE is considered as a probable human carcinogen. The epidemiologic evidence is based on various studies of lung cancer among truck drivers, bus drivers, shipyard workers, and railroad workers. Steenland et al. found a lifetime excess risk 10 times higher than the 1 per 1,000 excess risk allowed by Occupational Safety and Health Administration (OSHA) in setting regulations (136). Similar findings were also reported by Garshick et al., where they studied 31,135 male workers employed in the unionized U.S. trucking industry and found elevated risk of lung cancer in trucking industry workers (137). Several published critical reviews and epidemiologic meta-analyses (138-140) have reached to the conclusions supportive of DE exposure increasing lung cancer risk.
Traffic police
Traffic police often spend at least several hours per day driving or directing traffic in congested areas. These activities may result in exposure to carcinogenic airborne pollutants derived from motor vehicle exhaust, such as benzene, PAHs, and persistent organic pollutants, for example dioxin (141,142). Personal benzene exposures among police officers were three to five times higher than those observed among controls (143). PM is a major air pollution constituent that can induce inflammation, free radical formation, oxidative DNA damage, cytotoxicity, and mutagenesis within the respiratory tract (144). Although the frequency or extent of exposure to these agents amongst most law enforcement officers is not anticipated to be extensive, particularly with appropriate training and safety precautions. Potential exposure to these agents represents a growing concern within this occupation (145). Many studies have reached to conclusion supportive of exposure to traffic police, with an increased lung cancer risk.
Uranium mining
It is well established that high radon exposures increase the risk of lung cancer mortality in uranium miners. In a recently concluded joint cohort analysis of Czech, French, and Canadian uranium miners (employed in 1953 or later), they found that there is strong evidence for an increased risk of lung cancer mortality from low occupational radon exposures (146). Ramkissoon et al. described the risks of lung cancer by histological subtypes associated with exposure to radon decay products among the Ontario Uranium Miners cohort by using a retrospective cohort design. Authors found differences in the magnitude of the risks across four histological subtypes of lung carcinoma; the strongest association was noted for small cell lung carcinoma, followed by squamous cell, large cell, and lastly adenocarcinoma, which showed no significant association with exposure to radon decay products (147).
Existing policies and interventions
Although lung cancer is a global public health problem but many governments have not yet included lung cancer prevention in their health guidelines. Existing policies and guidelines related to environmental risk factors of lung cancer by different organizations and countries are summarized in Table 2.
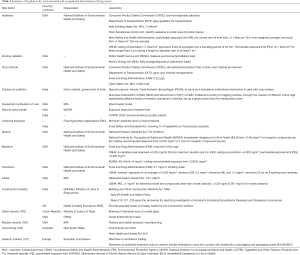
Full table
Asbestos (48)
Consumer Product Safety Commission (CPSC) has banned consumer patching compounds, free form of asbestos that could be respirable. Special guidelines have been set for marketing and transportation of this material by Department of Transportation (DOT). Maximum contamination level as per Safe Drinking Water Act has been fixed to 7 million fibers per liter for fibers longer than 10 µm. Toxic Substances Control Act Rules have been established for identifying, analyzing, and disposing of asbestos if asbestos is found in schools and public area.
Ionizing radiation (148)
Public Health Service Act (PHSA) and the Atomic Energy Act (AEA) directs authorities to measure environmental radiation levels, develop protective action guides, and provide assistance to the states for the safe storage and/or disposal of radioactive wastes.
Vinyl chloride (149)
CPSC has banned self-pressurized products intended for household use that contains vinyl chloride. DOT finds vinyl chloride as a hazardous material, and special requirements have been set for marking, labeling, and transporting this material. As per Clean Water Act Effluent Guidelines, it is listed as a toxic pollutant. Aerosol drug products have been withdrawn from the market and may not be compounded, because contained vinyl chloride was found to be unsafe or not effective. It is also banned from use in cosmetic aerosol products.
Indoor pollution
EPA has regulated household particulate emissions by giving wood heater model lines referred to as EPA-certified wood heaters (150). According to this model some guidelines should be followed to reduce household pollutions generated from coal combustion (as well as sulfur dioxide and oxides of nitrogen) levels (151).
Second-hand tobacco smoke
As per National Institute for Occupational Safety and Health (NIOSH), environmental tobacco smoke is considered a potential occupational carcinogen and exposure should be reduced to the lowest feasible concentration (152). In India, the Cigarettes and Other Tobacco Products Act 2003 prohibits smoking of tobacco in public places, except in special smoking zones in hotels, restaurants, airports and open spaces (153).
Chemical exposure
Pesticides maximum residue limits (MRLs) in food is guided by WHO guidelines on this issue. The Joint Food and Agriculture Organization of the United Nations (FAO)/WHO Meeting on Pesticide Residues (JMPR) evaluates those pesticides where contamination of food is likely (154). In India, the Food Safety and Standards Authority of India (FSSAI) was established under the Food Safety and Standards Act (155) to ensure safety of food materials from chemical contamination. These recommendations are important, considering the local needs of the states and research about specific crops, their diseases and insects.
Metals
Arsenic (156)
Arsenic compounds are identified as one of the 33 hazardous air pollutants that present the greatest threat to public health in urban areas as a part of Urban Air Toxics Strategy. Inorganic arsenic compounds are listed in the category of potential occupational carcinogens. A comprehensive set of guidelines has been established to prevent exposure to hazardous drugs in health-care settings by OSHA.
Beryllium (157)
Department of Energy (DOE) has established the Chronic Beryllium Disease Prevention Program to protect workers from excessive beryllium exposure and beryllium disease. Beryllium compounds are listed as hazardous air pollutants in EPA and Clean Air Act National Emission Standards for Hazardous Air Pollutants.
Chromium (158)
The U.S. EPA have established a maximum contaminant level of 0.1 mg/L for total chromium in drinking water. The FDA examines that the chromium concentration in bottled drinking water should not exceed 0.1 mg/L.
Nickel (159)
As per U.S. EPA, nickel compounds are listed as mobile-source air toxics for which regulations has to be developed. According to National Emission Standards for Hazardous Air Pollutants, nickel and its compounds come under the list of hazardous air pollutants. Nickel compounds are identified as one of the 33 hazardous air pollutants that present the greatest threat to public health in urban areas as per Urban Air Toxics Strategy.
Intervention for primary prevention
Primary prevention encompasses the elimination or reduction of recognized risk factors in susceptible populations and gives specific protection to prevent a disease. It is evident from various research studies that tobacco smoking is related to high incidence of lung cancer. Behaviour modification can help us to attain a fall in tobacco smoking, thereby reducing cases of lung cancer. There is a need to look into nontobacco risk factors, as lung cancer in nonsmokers is becoming a serious concern. This may be attributed to environmental and occupational risk factors. There is an increase in number of young lung cancer patients who are presenting to the hospital where target mutations are high in number in comparison to smokers. Effective primary preventive methods need to be explored as it is an important & cost-effective tool. Primary prevention for lung cancer involves identifying those risk factors associated with the development of lung cancer, as demonstrated by various epidemiological studies. Tobacco still plays the most important role but non-tobacco determinants must also be considered in the present scenario.
Communication
There is need for appropriate communication strategies to propagate the scientific and political messages to the society. The message that lung cancer has environmental and occupational causes is clear among the scientific community but not known to general population. Media has always played a key role in disseminating messages to a large population, to increase public perception about the disease, to educate people in general and to target groups for behavior modifications and to generate awareness about environmental and occupational determinants of lung cancer.
Supporting cancer campaigns and creating conditions to facilitate behavioral modification is essential, but it should be done with collective efforts.
Communication is the best means to raise awareness on the part of and to influence a large number of people. In addition, effective communication with national decision-makers requires new ideas and innovations to ensure wellbeing. Benefits should be measured not only in terms of productivity, but as health, social and environmental benefits.
Multi-sectoral approach
There is a need for establishing a multi-sectoral approach and partnerships to undertake primary prevention to deal with environmental and occupational determinants of lung cancer. Population or community-based interventions on the part of health and non-health sectors, such as environment, occupational, housing, industry and trade, and by community organizations, private enterprises are needed. Other key stake holders should be involved to reduce environmental and occupational exposures to ensure lung cancer prevention. The role of trade unions is also important to report the health issues of the employees to accomplish a transition to clean and safe industry. Additionally, media should be involved in the dissemination of information in a collaborative manner. Scientific research is supported by citizens through taxes and therefore there should be proper communication of purpose and results of research. The current difficulties in coordination of all stake holders involved in implementation, such as different Ministries: Health, Environment, Labour, Agriculture, Industry, Finance, etc., underlines the importance of a multisectoral approach to primary prevention of lung cancer.
Research
Research in the area of lung cancer prevention is important to recognize the areas where action is required to prevent environment and occupation related lung cancer. There is a need of strengthening of cross-sectional research on environment- and occupation-related risk factors attributing to lung cancer in nonsmokers. Population at risk should be identified, and link with timing of exposures to be established. Research on bio-genetic level to see genetic and epigenetic interactions with environmental risk factors to support the development of evidence-based preventive strategies; analyzing the molecular and cellular mechanisms, involved in the origin, development and progression of, environmentally-related lung cancer; encouraging analytical research—it includes risk assessment of environmental and occupational exposures; ensuring the rapid transfer of research outcomes to control measures for the benefit of all populations.
Challenges & opportunities
We can prevent lung cancer by effectual, attainable and affordable environmental and occupational interventions. There are many evidences based on research and knowledge about a particular risk factor that is related to environment & occupation for lung cancer, but action is not taken yet in many of the developing countries. Asbestos is still being used and very commonly in many developing countries including China and India. Some countries have guidelines to use tanning beds (160-163). In many countries there is no guideline on occupational hazards by their respective occupational health institutions, and no prevention policy of occupational lung cancer by the government. It has not been given high priority (164). Research to measure the magnitude of air pollution and its seasonal variations on lung cancer is needed (165). Exposure to environmental pollution varies across countries and cities, but availability of data is limited (166). There is need for strong mechanisms and methods to reduce exposure to substances that are used under unaware conditions, especially at developing world. There are studies that tell the environmental risk factors for lung cancer and its genetic susceptibility (167,168) but more research is needed. There is an increased risk of lung cancer due to environmental pollution (169-171). Policies to reduce the pollution level should be in priority. There should be monitoring and evaluation of carcinogenic risk factor data and it should be available for chemical and commercial industries, and testing should be performed before products are introduced in the market. More research is needed on incidence of lung cancer in non-smoker who are exposed to environmental pollution (172).
Conclusions
Trying different ways to prevent lung cancer is an effective way to reduce deaths and it would be highly cost-effective in terms of reduction of health-care costs. Primary prevention efforts need to be intensified to eliminate or minimize physical, chemical and biological exposures to known carcinogens and for the implementation of environmental interventions, including in work settings, to reduce the incidence of lung cancer, and the clinical, personal, economic and social burdens related to it.
Sincere efforts are required to address many carcinogens found in the environment, over which the individual has little control, and which require broad, public health driven action by public authorities at the national, regional and even international levels, engaging all stakeholders in a multi-sectoral, collaborative approach. These interventions have become important in view of increasing incidence of lung cancer in nonsmokers. There is a need for identification of the gaps and barriers, which will help to define a road map to better address the environmental and occupational determinants of lung cancer, and to develop a range of proposals for primary prevention to introduce environmental and occupational exposures into the global cancer agenda.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2108;68:394-424
- Ferlay J, Soerjomataram I, Ervik M, et al. editors. GLOBOCAN 2012: Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012 v1.0: IARC CancerBase No. 11. Lyon: International Agency for Research on Cancer, 2014.
- National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395-409. [Crossref] [PubMed]
- National Cancer Registry Program. Three-Year Report of Population Based Cancer Registries 2012-2014. [Online]. Delhi: Indian Council of Medical Research, 2016. Available online: http://www.ncdirindia.org/Download/Media_Release.pdf
- GLOBOCAN 2018. International Agency for Research on Cancer. WHO 2018. Available online: http://gco.iarc.fr/
- U.S. Department of Health and Human Services. The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 2014.
- U.S. Department of Health and Human Services. How Tobacco Smoke Causes Disease: What It Means to You. Atlanta: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 2010.
- World Health Organization. Global health risks: mortality and burden of disease attributable to selected major risks. Geneva: World Health Organization. 2009. Available online: http://www.who.int/iris/handle/10665/44203
- IARC. Household Use of Solid Fuels and High temperature Frying. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. WHO 2010.
- Öberg M, Woodward A, Jaakkola MS, et al. Global estimate of the burden of disease from second-hand smoke. Geneva: WHO, 2010, Vol. 18.
- Seow WJ, Hu W, Vermeulen R, et al. Household air pollution and lung cancer in China: a review of studies in Xuanwei. Chin J Cancer 2014;33:471-5. [PubMed]
- Mu L, Liu L, Niu R, et al. Indoor air pollution and risk of lung cancer among Chinese female non-smokers. Cancer Causes Control 2013;24:439-50. [Crossref] [PubMed]
- John LD. Self-care strategies used by patients with lung cancer to promote quality of life. Oncol Nurs Forum 2010;37:339-47. [Crossref] [PubMed]
- Islami F, Stoklosa M, Drope J, et al. Global and Regional Patterns of Tobacco Smoking and Tobacco Control Policies. Eur Urol Focus 2015;1:3-16. [Crossref] [PubMed]
- Mackay J, Amos A. Women and tobacco. Respirology 2003;8:123-30. [Crossref] [PubMed]
- Jindal SK, Behera D. Clinical spectrum of primary lung cancer: review of Chandigarh experience of 10 years. Lung India 1990;8:94-8.
- Behera D. Managing lung cancer in developing countries: difficulties and solutions. Indian J Chest Dis Allied Sci 2006;48:243-4. [PubMed]
- Rothman N, Wacholder S, Caporaso NE, et al. The use of common genetic polymorphisms to enhance the epidemiologic study of environmental carcinogens. Biochim Biophys Acta 2001;1471:C1-10. [PubMed]
- Lichtenstein P, Holm NV, Verkasalo PK, et al. Environmental and heritable factors in the causation of cancer analyses of cohorts of twins from Sweden, Denmark, and Finland. N Engl J Med 2000;343:78-85. [Crossref] [PubMed]
- Samet JM. Environmental causes of lung cancer: what do we know in 2003? Chest 2004;125:80S-3S. [Crossref] [PubMed]
- Prüss-Üstün A, Corvalán C. Preventing disease through healthy environments: Towards an estimate of the environmental burden of disease. WHO 2016.
- Chisolm JJ. Fouling one's own nest. Pediatrics 1978;62:614-7. [PubMed]
- Siemiatycki J, Richardson L, Straif K, et al. Listing occupational carcinogens. Environ Health Perspect 2004;112:1447-59. [Crossref] [PubMed]
- Thakur S, Dhiman M, Mantha AK, et al. APE1 modulates cellular responses to organophosphate pesticide- induced oxidative damage in non-small cell lung carcinoma A549 cells. Mol Cell Biochem 2018;441:201-216. [Crossref] [PubMed]
- An estimated 12.6 million deaths each year are attributable to unhealthy environments [Online]. WHO 2019. Available online: https://www.who.int/news-room/detail/15-03-2016-an-estimated-12-6-million-deaths-each-year-are-attributable-to-unhealthy-environments
- Bonner MR, Freeman LE, Hoppin JA, et al. Occupational Exposure to Pesticides and the Incidence of Lung Cancer in the Agricultural Health Study. Environ Health Perspect 2017;125:544-51. [Crossref] [PubMed]
- Alavanja MC, Dosemeci M, Samanic C, et al. Pesticides and lung cancer risk in the Agricultural Health Study cohort. Am J Epidemiol 2004;160:876-85. [Crossref] [PubMed]
- Walker NJ, Yoshizawa K, Miller RA, et al. Pulmonary lesions in female Harlan Sprague-Dawley rats following two-year oral treatment with dioxin-like compounds. Toxicol Pathol 2007;35:880-9. [Crossref] [PubMed]
- Walker NJ, Crockett PW, Nyska A, et al. Dose-additive carcinogenicity of a defined mixture of “dioxin-like compounds”. Environ Health Perspect 2005;113:43-8. [Crossref] [PubMed]
- Singh NP, Singh UP, Guan H, et al. Prenatal exposure to TCDD triggers significant modulation of microRNA expression profile in the thymus that affects consequent gene expression. PLoS One 2012;7:e45054. [Crossref] [PubMed]
- Boffetta P, Mundt KA, Adami HO, et al. TCDD and cancer: a critical review of epidemiologic studies. Crit Rev Toxicol 2011;41:622-36. [Crossref] [PubMed]
- Kogevinas M, Becher H, Benn T, et al. Cancer mortality in workers exposed to phenoxy herbicides, chlorophenols, and dioxins. An expanded and updated international cohort study. Am J Epidemiol 1997;145:1061-75. [Crossref] [PubMed]
- Burns CJ, Beard KK, Cartmill JB. Mortality in chemical workers potentially exposed to 2,4- dichlorophenoxyacetic acid (2,4-D) 1945-94: an update. Occup Environ Med 2001;58:24-30. [Crossref] [PubMed]
- Collins JJ, Bodner K, Aylward LL, et al. Mortality rates among workers exposed to dioxins in the manufacture of pentachlorophenol. J Occup Environ Med 2009;51:1212-9. [Crossref] [PubMed]
- Ruder AM, Yiin JH. Mortality of US pentachlorophenol production workers through 2005. Chemosphere 2011;83:851-61. [Crossref] [PubMed]
- Manuwald U, Velasco Garrido M, Berger J, et al. Mortality study of chemical workers exposed to dioxins: follow-up 23 years after chemical plant closure. Occup Environ Med 2012;69:636-42. [Crossref] [PubMed]
- Collins JJ, Bodner K, Aylward LL, et al. Mortality rates among trichlorophenol workers with exposure to 2, 3,7,8-tetrachlorodibenzo-p-dioxin. Am J Epidemiol 2009;170:501-6. [Crossref] [PubMed]
- Zendehdel R, Tayefeh-Rahimian R, Kabir A. Chronic exposure to chlorophenol related compounds in the pesticide production workplace and lung cancer: a meta-analysis. Asian Pac J Cancer Prev 2014;15:5149-53. [Crossref] [PubMed]
- Lee WJ, Baccarelli A, Tretiakova M, et al. Pesticide exposure and lung cancer mortality in Leningrad province in Russia. Environ Int 2006;32:412-6. [Crossref] [PubMed]
- EPA. US Toxics Release Inventory Public Data Release. EPA 260-R-01-001. 2001. Available online: http://www.epa.gov/tri/tridata/tri01/
- IARC. Diesel and Gasoline Engine Exhausts and Some Nitroarenes. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Lyon: IARC, 1989, Vol. 46.
- Cohen AJ. Outdoor air pollution and lung cancer. Environ Health Perspect 2000;108:743-50. [PubMed]
- Lelieveld J, Evans JS, Fnais M, et al. The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 2015;525:367-71. [Crossref] [PubMed]
- IARC. Agents Classified by the IARC Monographs, Volumes 1–123 [Online]. WHO. Available online: https://monographs.iarc.fr/agents-classified-by-the-iarc/
- Raspanti GA, Hashibe M, Siwakoti B, et al. Household air pollution and lung cancer risk among never-smokers in Nepal. Environ Res 2016;147:141-5. [Crossref] [PubMed]
- Sapkota A, Gajalakshmi V, Jetly DH, et al. Indoor air pollution from solid fuels and risk of hypopharyngeal/laryngeal and lung cancers: a multicentric case-control study from India. Int J Epidemiol 2008;37:321-8. [Crossref] [PubMed]
- He F, Qi Q, Li X, et al. Association of Indoor Air Pollution, Single Nucleotide Polymorphism of HIF-1α Gene with Susceptibility to Lung Cancer in Han Population in Fujian Province. Zhongguo Fei Ai Za Zhi 2017;20:149-56. [PubMed]
- National Toxicology Program: 14th Report on Carcinogens [Online]. National Toxicology Program (NTP). 2019. Available online: https://ntp.niehs.nih.gov/pubhealth/roc/index-1.html
- Butler KM, Rayens MK, Wiggins AT, et al. Association of Smoking in the Home With Lung Cancer Worry, Perceived Risk, and Synergistic Risk. Oncol Nurs Forum 2017;44:E55-63. [PubMed]
- Sun YQ, Chen Y, Langhammer A, et al. Passive smoking in relation to lung cancer incidence and histologic types in Norwegian adults: the HUNT study. Eur Respir J 2017;50. [Crossref] [PubMed]
- Avino P, Scungio M, Stabile L, et al. Second-hand aerosol from tobacco and electronic cigarettes: Evaluation of the smoker emission rates and doses and lung cancer risk of passive smokers and vapers. Sci Total Environ 2018;642:137-47. [Crossref] [PubMed]
- Rapiti E, Jindal SK, Gupta D, et al. Passive smoking and lung cancer in Chandigarh, India. Lung Cancer 1999;23:183-9. [Crossref] [PubMed]
- Das A, Krishnamurthy A, Ramshankar V, et al. The increasing challenge of never smokers with adenocarcinoma lung: Need to look beyond tobacco exposure. Indian J Cancer 2017;54:172-7. [Crossref] [PubMed]
- US Environmental Protection Agency. Integrated Risk Information System (IRIS) on Asbestos. Washington, DC: National Center for Environmental Assessment. Office of Research and Development. Available online: https://cfpub.epa.gov/ncea/iris/iris_documents/documents/subst/1026_summary.pdf
- Agency for Toxic Substances and Disease Registry (ATSDR). Toxicological Profile for Asbestos. Atlanta, GA: Public Health Service, U.S. Department of Health and Human Services. 2001. Available online: http://www.atsdr.cdc.gov/ToxProfiles/tp61.pdf
- National Toxicology Program (NTP). Report on Carcinogens, Thirteenth Edition. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service. 2014. Available online: http://ntp.niehs.nih.gov/pubhealth/roc/roc13/4
- Asbestos [Online]. World Health Organization 2019. Available online: https://www.who.int/ipcs/assessment/public_health/asbestos/en/
- Selikoff IJ, Lee DHK. Asbestos and Disease. Cambridge, MA: Academic Press, 1978.
- Järvholm B, Englund A, Albin M. Pleural mesothelioma in Sweden: an analysis of the incidence according to the use of asbestos. Occup Environ Med 1999;56:110-3. [Crossref] [PubMed]
- Järvholm B, Aström E. The Risk of Lung Cancer After Cessation of Asbestos Exposure in Construction Workers Using Pleural Malignant Mesothelioma as a Marker of Exposure. J Occup Environ Med 2014;56:1297-301. [Crossref] [PubMed]
- Kagan E. Asbestos-induced mesothelioma: is fiber biopersistence really a critical factor? Am J Pathol 2013;183:1378-81. [Crossref] [PubMed]
- Kazan-Allen L. Killing the future—asbestos use in Asia. International Ban Asbestos Secretariat, UK. 2007. Available online: http://www.ibasecretariat.org/ktf_web_fin.pdf
- Lubin JH, Moore LE, Fraumeni JF Jr, et al. Respiratory cancer and inhaled inorganic arsenic in copper smelter workers: a linear relationship with cumulative exposure that increases with concentration. Environ Health Perspect 2008;116:1661-5. [Crossref] [PubMed]
- Cantor KP, Lubin JH. Arsenic, internal cancers, and issues in inference from studies of low level exposures in human populations. Toxicol Appl Pharmacol 2007;222:252-7. [Crossref] [PubMed]
- Mosquin PL, Rothman KJ. Reanalysis of Reported Associations of Beryllium and Lung Cancer in a Large Occupational Cohort. J Occup Environ Med 2017;59:274-81. [Crossref] [PubMed]
- Levy PS, Roth HD, Deubner DC. Exposure to beryllium and occurrence of lung cancer: findings from a cox proportional hazards analysis of data from a retrospective cohort mortality study. J Occup Environ Med 2009;51:480-6. [Crossref] [PubMed]
- Schubauer-Berigan MK, Deddens JA, Couch JR, et al. Risk of lung cancer associated with quantitative beryllium exposure metrics within an occupational cohort. Occup Environ Med 2011;68:354-60. [Crossref] [PubMed]
- Agency for Toxic Substances and Disease Registry. Toxic Substances Portal - Beryllium. Atlanta, GA: Centers for Disease Control and Prevention, 2014.
- International Agency for Research on Cancer. Beryllium and Beryllium Compounds, IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 100C. Lyon, France: World Health Organization, 2012.
- Sarlinova M, Majerova L, Matakova T, et al. Polymorphisms of DNA repair genes and lung cancer in chromium exposure. Adv Exp Med Biol 2015;833:1-8. [PubMed]
- Gibb HJ, Lees PS, Pinsky PF, et al. Lung cancer among workers in chromium chemical production. Am J Ind Med 2000;38:115-26. [Crossref] [PubMed]
- U.S. Environmental Protection Agency. Integrated Risk Information System (IRIS) on Nickel, Soluble Salts. Washington, DC: National Center for Environmental Assessment, Office of Research and Development, 1999.
- Chiou YH, Wong RH, Chao MR, et al. Nickel accumulation in lung tissues is associated with increased risk of p53 mutation in lung cancer patients. Environ Mol Mutagen 2014;55:624-32. [Crossref] [PubMed]
- Grimsrud TK, Berge SR, Haldorsen T, et al. Exposure to different forms of nickel and risk of lung cancer. Am J Epidemiol 2002;156:1123-32. [Crossref] [PubMed]
- Bernacki EJ, Parsons GE, Sunderman FW Jr. Investigation of exposure to nickel and lung cancer mortality: case control study at aircraft engine factory. Ann Clin Lab Sci 1978;8:190-4. [PubMed]
- Mastrangelo G, Fedeli U, Fadda E, et al. Lung cancer risk in workers exposed to poly (vinyl chloride) dust: a nested case-referent study. Occup Environ Med 2003;60:423-8. [Crossref] [PubMed]
- Scélo G, Constantinescu V, Csiki I, et al. Occupational exposure to vinyl chloride, acrylonitrile and styrene and lung cancer risk (europe). Cancer Causes Control 2004;15:445-52. [Crossref] [PubMed]
- ATSDR. Toxicological Profile for Vinyl Chloride. Agency for Toxic Substances and Disease Registry. 2006. Available online: http://www.atsdr.cdc.gov/toxprofiles/tp20.pdf
- HSDB. Hazardous Substances Data Bank. National Library of Medicine. search on CAS number. Last accessed: 10/22/09. 2009. Available online: http://toxnet.nlm.nih.gov/cgi-bin/sis/htmlgen?HSDB
- Kielhorn J, Melber C, Wahnschaffe U, et al. Vinyl chloride: still a cause for concern. Environ Health Perspect 2000;108:579-88. [Crossref] [PubMed]
- Field RW, Steck DJ, Smith BJ, et al. Residential radon gas exposure and lung cancer: the Iowa Radon Lung Cancer Study. Am J Epidemiol 2000;151:1091-102. [Crossref] [PubMed]
- Alavanja MC, Lubin JH, Mahaffey JA, et al. Residential radon exposure and risk of lung cancer in Missouri. Am J Public Health 1999;89:1042-8. [Crossref] [PubMed]
- Krewski D, Lubin JH, Zielinski JM, et al. A combined analysis of North American case-control studies of residential radon and lung cancer. J Toxicol Environ Health A 2006;69:533-97. [Crossref] [PubMed]
- Darby S, Hill D, Deo H, et al. Residential radon and lung cancer--detailed results of a collaborative analysis of individual data on 7148 persons with lung cancer and 14,208 persons without lung cancer from 13 epidemiologic studies in Europe. Scand J Work Environ Health 2006;32:1-83. [PubMed]
- Tirmarche M, Baysson H, Telle-Lamberton M. Uranium exposure and cancer risk: a review of epidemiological studies. Rev Epidemiol Sante Publique 2004;52:81-90. [Crossref] [PubMed]
- Lindsay JP, Stavraky KM, Howe GR. The Canadian Labour Force Ten Percent Sample Study. Cancer mortality among men, 1965-1979. J Occup Med 1993;35:408-14. [PubMed]
- Office of Health and Environmental Assessment EPAE. Respiratory health effects of passive smoking: lung cancer and other disorders. Washington, DC: EPA, 1992.
- Siegel M, Skeer M. Exposure to secondhand smoke and excess lung cancer mortality risk among workers in the "5 B's": bars, bowling alleys, billiard halls, betting establishments, and bingo parlours. Tob Control 2003;12:333-8. [Crossref] [PubMed]
- LeMasters G, Lockey JE, Hilbert TJ, et al. A 30-year mortality and respiratory morbidity study of refractory ceramic fiber workers. Inhal Toxicol 2017;29:462-70. [Crossref] [PubMed]
- Meijers JM, Swaen GM, Slangen JJ. Mortality and lung cancer in ceramic workers in The Netherlands: preliminary results. Am J Ind Med 1996;30:26-30. [Crossref] [PubMed]
- Forastiere F, Lagorio S, Michelozzi P, et al. Silica, silicosis and lung cancer among ceramic workers: a case- referent study. Am J Ind Med 1986;10:363-70. [Crossref] [PubMed]
- Singh A, Kamal R, Ahamed I, et al. PAH exposure-associated lung cancer: an updated meta-analysis. Occup Med (Lond) 2018;68:255-61. [Crossref] [PubMed]
- Berger J, Manz A. Cancer of the stomach and the colon-rectum among workers in a coke gas plant. Am J Ind Med 1992;22:825-34. [Crossref] [PubMed]
- Martin JC, Imbernon E, Goldberg M, et al. Occupational risk factors for lung cancer in the French electricity and gas industry: a case-control survey nested in a cohort of active employees. Am J Epidemiol 2000;151:902-12. [Crossref] [PubMed]
- Burkhart G, Schulte PA, Robinson C, et al. Job tasks, potential exposures, and health risks of laborers employed in the construction industry. Am J Ind Med 1993;24:413-25. [Crossref] [PubMed]
- Dement JM, Ringen K, Welch LS, et al. Mortality of older construction and craft workers employed at Department of Energy (DOE] nuclear sites. Am J Ind Med 2009;52:671-82. [Crossref] [PubMed]
- Dong W, Vaughan P, Sullivan K, et al. Mortality study of construction workers in the UK. Int J Epidemiol 1995;24:750-7. [Crossref] [PubMed]
- Hall NE, Rosenman KD. Cancer by industry: analysis of a population-based cancer registry with an emphasis on blue-collar workers. Am J Ind Med 1991;19:145-59. [Crossref] [PubMed]
- Koskinen K, Pukkala E, Martikainen R, et al. Different measures of asbestos exposure in estimating risk of lung cancer and mesothelioma among construction workers. J Occup Environ Med 2002;44:1190-6. [Crossref] [PubMed]
- Robinson C, Stern F, Halperin W, et al. Assessment of mortality in the construction industry in the United States, 1984–1986. Am J Ind Med 1995;28:49-70. [Crossref] [PubMed]
- Stern F, Schulte P, Sweeney MH, et al. Proportionate mortality among construction laborers. Am J Ind Med 1995;27:485-509. [Crossref] [PubMed]
- Stocks SJ, McNamee R, Carder M, et al. The incidence of medically reported work-related ill health in the UK construction industry. Occup Environ Med 2010;67:574-6. [Crossref] [PubMed]
- Thuret A, Geoffroy-Perez B, Luce D, et al. A 26-year cohort mortality study of French construction workers aged 20 to 64 years. J Occup Environ Med 2007;49:546-56. [Crossref] [PubMed]
- Veglia F, Vineis P, Overvad K, et al. Occupational exposures, environmental tobacco smoke, and lung cancer. Epidemiology 2007;18:769-75. [Crossref] [PubMed]
- Wang E, Dement JM, Lipscomb H. Mortality among North Carolina construction workers, 1988–1994. Appl Occup Environ Hyg 1999;14:45-58. [Crossref] [PubMed]
- Arndt V, Rothenbacher D, Daniel U, et al. All-cause and cause specific mortality in a cohort of 20 000 construction workers; results from a 10 year follow up. Occup Environ Med 2004;61:419-25. [Crossref] [PubMed]
- d’Errico A, Mamo C, Tomaino A, et al. Mortality of a cohort of road construction and maintenance workers with work disability compensation. Med Lav 2002;93:519-26. [PubMed]
- Engholm G, Englund A. Mortality and cancer incidence in various groups of construction workers. Occup Med 1995;10:453-81. [PubMed]
- Sun J, Kubota H, Hisanaga N, et al. Mortality among Japanese construction workers in Mie Prefecture. Occup Environ Med 2002;59:512-6. [Crossref] [PubMed]
- Finkelstein MM, Verma DK. Mortality among Ontario members of the International Union of Bricklayers and Allied Craftworkers. Am J Ind Med 2005;47:4-9. [Crossref] [PubMed]
- Minder CE, Beer-Porizek V. Cancer mortality of Swiss men by occupation, 1979–1982. Scand J Work Environ Health 1992;18 Suppl 3:1-27. [PubMed]
- Salg J, Alterman T. A proportionate mortality study of bricklayers and allied craftworkers. Am J Ind Med 2005;47:10-9. [Crossref] [PubMed]
- Stern F, Lehman E, Ruder A. Mortality among unionized construction plasterers and cement masons. Am J Ind Med 2001;39:373-88. [Crossref] [PubMed]
- Consonni D, De Matteis S, Pesatori AC, et al. Lung cancer risk among bricklayers in a pooled analysis of case–control studies. Int J Cancer 2015;136:360-71. [Crossref] [PubMed]
- Dement J, Pompeii L, Lipkus IM, et al. Cancer incidence among Union carpenters in New Jersey. J Occup Environ Med 2003;45:1059-67. [Crossref] [PubMed]
- Robinson CF, Petersen M, Sieber WK, et al. Mortality of Carpenters’ Union members employed in the U.S. construction or wood products industries, 1987–1990. Am J Ind Med 1996;30:674-94. [Crossref] [PubMed]
- Steenland K, Palu S. Cohort mortality study of 57,000 painters and other union members: a 15 year update. Occup Environ Med 1999;56:315-21. [Crossref] [PubMed]
- Stern F, Haring-Sweeney M. Proportionate mortality among unionized construction operating engineers. Am J Ind Med 1997;32:51-65. [Crossref] [PubMed]
- Wong O, Foliart D, Trent LS. A case-control study of lung cancer in a cohort of workers potentially exposed to slag wool fibres. Br J Ind Med 1991;48:818-24. [PubMed]
- Sankila R, Karjalainen S, Pukkala E, et al. Cancer risk among glass factory workers: an excess of lung cancer? Br J Ind Med 1990;47:815-8. [PubMed]
- Wingren G, Englander V. Mortality and cancer morbidity in a cohort of Swedish glassworkers. Int Arch Occup Environ Health 1990;62:253-7. [Crossref] [PubMed]
- Shannon H, Muir A, Haines T, et al. Mortality and cancer incidence in Ontario glass fiber workers. Occup Med (Lond) 2005;55:528-34. [Crossref] [PubMed]
- Consonni D, De Matteis S, Pesatori AC, et al. Increased lung cancer risk among bricklayers in an Italian population-based case control study. Am J Ind Med 2012;55:423-8. [Crossref] [PubMed]
- IARC. IARC Monographs on the evaluation of carcinogenic risks to humans, Vol. 47. Some organic solvents, resin monomers and related compounds, pigments and occupational exposures in paint manufacture and painting. Lyon: IARC (International Agency for Research on Cancer), 1989.
- Straif K, Baan R, Grosse Y, et al. Carcinogenicity of shift-work, painting, and fire-fighting. Lancet Oncol 2007;8:1065-6. [Crossref] [PubMed]
- Ramanakumar AV, Parent MÉ, Richardson L, et al. Exposures in painting-related occupations and risk of lung cancer among men: results from two case-control studies in Montreal. Occup Environ Med 2011;68:44-51. [Crossref] [PubMed]
- IARC. Occupational exposures in the rubber manufacturing industry. In: Chemical Agents and Related Occupations. Lyon, France: International Agency for Research on Cancer, 2012.
- IARC. The Rubber Industry. Lyon, France: International Agency for Research on Cancer, 1982.
- Dosemeci M, Rothman N, Yin SN, et al. Validation of benzene exposure assessment. Ann N Y Acad Sci 1997;837:114-21. [Crossref] [PubMed]
- Dollard GJ, Dore CJ, Jenkin ME. Ambient concentrations of 1,3-butadiene in the UK. Chem Biol Interact 2001;135-136:177-206. [Crossref] [PubMed]
- Boniol M, Koechlin A, Boyle P. Meta-analysis of occupational exposures in the rubber manufacturing industry and risk of cancer. Int J Epidemiol 2017;46:1940-7. [Crossref] [PubMed]
- International Agency for Research on Cancer. Silica, some silicates, coal dust, and para-aramid fibrils. Monograph 68. Evaluation of carcinogenic risks to humans. Lyon, France: International Agency for Research on Cancer, 1997.
- Steenland K, Sanderson W. Lung cancer among industrial sand workers exposed to crystalline silica. Am J Epidemiol 2001;153:695-703. [Crossref] [PubMed]
- Güngen AC, Aydemir Y, Çoban H, et al. Lung cancer in patients diagnosed with silicosis should be investigated. Respir Med Case Rep 2016;18:93-5. [Crossref] [PubMed]
- Satpathy SR, Jala VR, Bodduluri SR, et al. Crystalline silica-induced leukotriene B4-dependent inflammation promotes lung tumour growth. Nat Commun 2015;6:7064. [Crossref] [PubMed]
- Steenland K, Deddens J, Stayner L. Diesel exhaust and lung cancer in the trucking industry: exposure- response analyses and risk assessment. Am J Ind Med 1998;34:220-8. [Crossref] [PubMed]
- Garshick E, Laden F, Hart JE, et al. Lung cancer and vehicle exhaust in trucking industry workers. Environ Health Perspect 2008;116:1327-32. [Crossref] [PubMed]
- Lipsett M, Campleman S. Occupational exposure to diesel exhaust and lung cancer: a meta-analysis. Am J Public Health 1999;89:1009-17. [Crossref] [PubMed]
- Lloyd AC, Cackette TA. Diesel engines: environmental impact and control. J Air Waste Manag Assoc 2001;51:809-47. [Crossref] [PubMed]
- Wichmann HE. Diesel exhaust particles. Inhal Toxicol 2007;19 Suppl 1:241-4. [Crossref] [PubMed]
- Forastiere F, Perucci CA, Di Pietro A, et al. Mortality among urban policemen in Rome. Am J Ind Med 1994;26:785-98. [Crossref] [PubMed]
- Ritter L, Soloman KR, Forget J. Persistent Organic Pollutants: An assessment report on DDTAldrin-Dieldrin-Endrin-Chlordane Heptachlor-Hexachlorobenzene Mirex-Toxaphene polychlorinated biphenyls dioxins furans. The International Program on Chemical Safety (IPCS). 2007. Available online: http://www.whoint/ipcs/en/ on November 27 2012
- Pilidis GA, Karakitsios SP, Kassomenos PA, et al. Measurements of benzene and formaldehyde in a medium sized urban environment. Indoor/outdoor health risk implications on special population groups. Environ Monit Assess 2009;150:285-94. [Crossref] [PubMed]
- Valavanidis A, Fiotakis K, Vlachogianni T. Airborne particulate matter and human health: toxicological assessment and importance of size and composition of particles for oxidative damage and carcinogenic mechanisms. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 2008;26:339-62. [Crossref] [PubMed]
- Thrasher DL, Von Derau K, Burgess J. Health effects from reported exposure to methamphetamine labs: A poison center-based study. J Med Toxicol 2009;5:200-4. [Crossref] [PubMed]
- Lane RSD, Tomášek L, Zablotska LB, et al. Low radon exposures and lung cancer risk: joint analysis of the Czech, French, and Beaverlodge cohorts of uranium miners. Int Arch Occup Environ Health 2019. [Epub ahead of print]. [Crossref] [PubMed]
- Ramkissoon A, Navaranjan G, Berriault C, et al. Histopathologic Analysis of Lung Cancer Incidence Associated with Radon Exposure among Ontario Uranium Miners. Int J Environ Res Public Health 2018;15. [Crossref] [PubMed]
- Radiation Protection at EPA [Online]. 2011. U.S. Environmental Protection Agency. Available online: https://www.epa.gov/sites/production/files/2015-05/documents/402-b-00-001.pdf
- National Toxicology Program. Vinyl Halides (Selected), Report on Carcinogens, Fourteenth Edition. Triangle Park, NC: National Institute of Environmental Health and Safety, 2016. Available online: https://ntp.niehs.nih.gov/pubhealth/roc/index-1.html
- Particulate Matter (PM) Implementation Regulatory Actions [Online]. US Environmental Protection Agency. Available online: https://www.epa.gov/pm-pollution/particulate-matter-pm-implementation-regulatory-actions
- Centers for Disease Control and Prevention and U.S. Department of Housing and Urban Development. Chapter 5: Indoor Air Pollutants and Toxic Materials. [Online]. Atlanta: US Department of Health and Human Services; 2006. Available online: https://www.cdc.gov/nceh/publications/books/housing/housing_ref_manual_2012.pdf
- National Toxicology Program. Tobacco-Related Exposures, Report on Carcinogens, Fourteenth Edition [Online]. Triangle Park, NC: National Institute of Environmental Health and Safety, 2016. Available online: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/tobaccorelatedexposures.pdf
- Government of India. The Cigarettes and Other Tobacco Products (Prohibition of Advertisement and Regulation of Trade and Commerce, Production, Supply and Distribution) Act, 2003 [Online]. 2003. Available online: https://indiacode.nic.in/bitstream/123456789/2053/1/200334.pdf
- FAO/WHO (2008). Second session of the FAO/WHO meeting on pesticide management and 4th session of the FAO panel of experts on pesticide management [Online]. Geneva, World Health Organization. Available online: http://www.fao.org/fileadmin/templates/agphome/documents/Pests_Pesticides/Code/Recom mendations08_01.pdf
- Bhushan C, Bhardwaj A, Misra SS. State of Pesticide Regulations in India. New Delhi: Centre for Science and Environment, 2013. Available online: https://www.cseindia.org/state-of-pesticide-regulations-in-india-7611
- National Toxicology Program. Arsenic and Inorganic Arsenic Compounds, Report on Carcinogens, Fourteenth Edition [Online]. Research Triangle Park, NC: U.S. Department of Health and Human Services, Public Health Service, 2016. Available online: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/arsenic.pdf
- National Toxicology Program. Beryllium and Beryllium Compounds, Report on Carcinogens, Fourteenth Edition [Online]. Triangle Park, NC: National Institute of Environmental Health and Safety, 2016. Available online: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/beryllium.pdf
- National Toxicology Program. Chromium Hexavalent Compounds, Report on Carcinogens, Fourteenth Edition [Online]. Triangle Park, NC: National Institute of Environmental Health and Safety, 2016. Available online: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/chromiumhexavalentcompounds.pdf
- National Toxicology Program. Nickel Compounds and Metallic Nickel, Report on Carcinogens, Fourteenth Edition [Online]. Triangle Park, NC: National Institute of Environmental Health and Safety, 2016. Available online: https://ntp.niehs.nih.gov/ntp/roc/content/profiles/nickel.pdf
- Choy CC, Cartmel B, Clare RA, et al. Compliance with indoor tanning bans for minors among businesses in the USA. Transl Behav Med 2017;7:637-44. [Crossref] [PubMed]
- From Australia to Brazil: sun worshippers beware. Bull World Health Organ 2009;87:574-5. [Crossref] [PubMed]
- Day AK, Coups EJ, Manne SL, et al. Recall of indoor tanning salon warnings and safety guidelines among a national sample of tanners. Transl Behav Med 2016;6:622-7. [Crossref] [PubMed]
- Mogensen M, Jemec GB. The potential carcinogenic risk of tanning beds: clinical guidelines and patient safety advice. Cancer Manag Res 2010;2:277-82. [Crossref] [PubMed]
- Jilcha K, Kitaw D. A Literature Review on Global Occupational Safety and Health Practice & Accidents Severity. International Journal for Quality Research 2016;10:279-310.
- Parascandola M. Ambient air pollution and lung cancer in Poland: research findings and gaps. J Health Inequal 2018;4:3-8. [Crossref]
- Jedrychowski W, Maugeri U, Bianchi I. Environmental pollution in central and eastern European countries: a basis for cancer epidemiology. Rev Environ Health 1997;12:1-23. [Crossref] [PubMed]
- Bennett WP, Alavanja MC, Blomeke B, et al. Environmental tobacco smoke, genetic susceptibility, and risk of lung cancer in never-smoking women. J Natl Cancer Inst 1999;91:2009-14. [Crossref] [PubMed]
- Yang IA, Holloway JW, Fong KM. Genetic susceptibility to lung cancer and co-morbidities. J Thorac Dis 2013;5 Suppl 5:S454-62. [PubMed]
- Moore JX, Akinyemiju T, Wang HE. Pollution and regional variations of lung cancer mortality in the United States. Cancer Epidemiol 2017;49:118-27. [Crossref] [PubMed]
- Jedrychowski W, Becher H, Wahrendorf J, et al. A case-control study of lung cancer with special reference to the effect of air pollution in Poland. J Epidemiol Community Health 1990;44:114-20. [Crossref] [PubMed]
- Huang F, Pan B, Wu J, et al. Relationship between exposure to PM2.5 and lung cancer incidence and mortality: A meta-analysis. Oncotarget 2017;8:43322-31. [PubMed]
- Lamichhane DK, Kim HC, Choi CM, et al. Lung Cancer Risk and Residential Exposure to Air Pollution: A Korean Population-Based Case-Control Study. Yonsei Med J 2017;58:1111-8. [Crossref] [PubMed]


