Real-world insights into multidisciplinary management and outcomes of stage III (N2–3) non-small cell lung cancer
Highlight box
Key findings
• Novel neoadjuvant therapies improved pathological response and survival in stage III (N2–3) non-small cell lung cancer (NSCLC).
• Immunotherapy-chemotherapy (IO-chemo) achieved the highest surgical proportion and superior pathological complete response/major pathological response rates.
• Median survival of the entire cohort reached 67.2 months, and outcomes after surgery were better.
• Maintenance therapy after local treatment significantly improved event-free and overall survival.
• Even patients with N3 disease derived survival benefit from surgery following effective neoadjuvant therapy.
What is known and what is new?
• Treatment of stage III (N2–3) NSCLC is challenging due to its heterogeneity, and evidence for neoadjuvant immunotherapy or targeted therapy in this population is limited.
• This real-world study demonstrated that IO-chemo and targeted therapy significantly improved pathological response and long-term survival compared with chemotherapy alone, and that selected N2–3 patients may still benefit from surgery after effective neoadjuvant therapy.
What is the implication, and what should change now?
• Integration of immunotherapy or targeted therapy into multidisciplinary management should be encouraged for patients with stage III (N2–3) NSCLC.
• Surgery should be considered for selected N2–3 patients who respond well to neoadjuvant treatment.
• Maintenance therapy should be included as part of standard post-local treatment care to optimize long-term outcomes.
Introduction
Non-small cell lung cancer (NSCLC) remains one of the leading causes of cancer incidence and mortality worldwide (1,2). Approximately 20–35% of patients are diagnosed with stage III disease (3). Most patients with stage III disease present with loco-regional lymph node involvement at initial diagnosis (4). The substantial clinical and molecular heterogeneity in this population complicates subsequent treatment and makes curative resection particularly challenging (5). As a result, therapeutic strategies vary widely across institutions and remain a matter of ongoing clinical debate.
In current practice, multidisciplinary treatment (MDT) approaches for stage III NSCLC include surgery-centered and radiotherapy-centered strategies (5,6). With the rapid development and widespread adoption of novel systemic therapies, neoadjuvant treatment options have expanded considerably (7). Immunotherapy- and targeted therapy-based regimens have demonstrated higher objective response rates than traditional chemotherapy and improved prospects for long-term survival (8-10). However, real-world evidence remains limited regarding how these regimens are applied in patients with stage III (N2–3) disease, how different neoadjuvant strategies influence pathological response and survival outcomes, and how subsequent treatment pathways are determined.
To address these gaps, we conducted a real-world study of patients with stage III (N2–3) NSCLC treated at a high-volume cancer center. We systematically assessed neoadjuvant strategies, subsequent local therapies, and survival outcomes. We aimed to provide evidence-based insight to guide individualized treatment decision for this challenging patient population. We present this article in accordance with the TREND reporting checklist (available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-aw-1230/rc).
Methods
Patient selection
Patients with lung tumors treated at our center between 2009 and 2024 were retrospectively identified from the electronic medical record system. Only patients who underwent mediastinoscopy or endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) for pathological nodal staging were eligible for inclusion. Exclusion criteria were as follows: (I) small cell lung cancer (SCLC) patients; (II) presence of distant metastasis (M1 disease); (III) N0 or N1 disease; (IV) no receipt of neoadjuvant therapy; and (V) prior tumor history. Participation in clinical trials was not an exclusion criterion. The patient selection flowchart is presented in Figure 1. For analytical purposes, patients were first stratified by driver gene mutation status into three neoadjuvant treatment groups: neoadjuvant immuno-chemotherapy (IO-chemo), targeted therapy, and chemotherapy. Following neoadjuvant therapy, patients were further categorized into three groups according to subsequent local managements: surgery, radiotherapy, or continuation of systemic therapy.

Ethical statement
This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments (11). The study was approved by the Ethics Committee of Sun Yat-sen University Cancer Center (No. B2025-452-01) and individual consent for this retrospective analysis was waived.
Data collection
Collected data included demographic and clinical variables such as age, sex, smoking history (yes for current or former smokers and no for never smokers), Eastern Cooperative Oncology Group performance status (ECOG PS), date of pathological diagnosis, histopathological findings from biopsy specimens, pretreatment clinical staging, tumor location, programmed cell death ligand 1 (PD-L1) expression level, details of neoadjuvant therapy, pre- and post-treatment imaging, date of surgery or radiotherapy initiation, pathological results of surgical specimens, pathological response, maintenance therapy, and follow-up information.
Maintenance therapy was defined as continued systemic treatment administered after completion of neoadjuvant therapy and surgery or radiotherapy, including (I) tyrosine kinase inhibitors (TKIs) (such as osimertinib, alectinib, and others); (II) immune checkpoint inhibitors used as continuation or consolidation (e.g., nivolumab, pembrolizumab, and others); and (III) chemotherapy-based regimens or anti-angiogenic combinations where applicable. Maintenance therapies varied according to tumor biology, treatment tolerability, and MDT recommendations, and were continued until disease progression or unacceptable toxicity. Clinical and pathological staging were based on the 8th edition Tumor-Node-Metastasis (TNM) staging system (12). Clinical nodal status (cN) was determined using mediastinoscopy or EBUS-TBNA. Pathological response was evaluated on resected specimens (performed by a senior pathologist: J.T.J.) (13). Pathological complete response (pCR) was defined as the absence of any residual viable tumor cells in both the primary tumor and sampled lymph nodes. Major pathological response (MPR) was defined as ≤10% residual viable tumor cells in the primary tumor bed, irrespective of nodal status.
Neoadjuvant therapy
In this study, neoadjuvant chemotherapy consisted of platinum-based doublets, including pemetrexed-platinum, taxane-platinum, and gemcitabine-platinum, administered for 2–4 cycles. IO-chemo regimens combined a platinum-based doublet with an immune checkpoint inhibitor (including pembrolizumab, nivolumab, atezolizumab, and others) for 2–4 cycles. PD-L1 expression was retrospectively collected when available. Targeted therapy regimen was selected according to driver alterations and included epidermal growth factor receptor (EGFR) TKIs (gefitinib, icotinib, osimertinib, and others), anaplastic lymphoma kinase (ALK) inhibitors (crizotinib, alectinib), and a small number of other genotype-matched agents, given for approximately 8–12 weeks before re-evaluation.
Study endpoints
The primary endpoints were overall survival (OS) and event-free survival (EFS). OS was defined as the time from pathological diagnosis to death from any cause, while EFS was defined as the time from pathological diagnosis to disease progression or death from any cause, whichever occurred first. Secondary endpoints included surgical conversion rate, MPR rate, and pCR rate.
Statistical analysis
All statistical analyses were performed using R software (version 4.4.2). Descriptive statistics were used to summarize baseline clinicopathological characteristics. Continuous variables were reported as medians with ranges, and categorical variables were presented as number with percentages. Group comparisons of categorical variables were performed using Pearson’s chi-square test or Fisher’s exact test, as appropriate. Survival curves were generated using the Kaplan-Meier method, and differences were assessed with the log-rank test (14). Variables with P<0.05 in univariate analysis were entered into the multivariate Cox regression model. A two-sided P value <0.05 was considered statistically significant.
Results
Baseline characteristics
Between 2009 and 2024, a total of 116,368 patients diagnosed with lung tumors at our center were screened for eligibility. After applying the inclusion and exclusion criteria, 243 patients were eligible for this study, including 158 patients who underwent EBUS-TBNA and 85 patients who received mediastinoscopy lymph node examination. The baseline clinicopathological characteristics of the study population are summarized in Table 1. The median age was 60 years (range, 33–78 years). Of these, 169 patients (69.5%) were male, and 125 (51.4%) had a smoking history. Adenocarcinoma was the most common pathological type (74.1%). The right lower lobe was the most common primary tumor site, observed in 91 cases (37.4%). 175 patients (72.0%) were pathologically staged as pN2, while 68 patients (28.0%) were staged as pN3. At initial diagnosis, stage IIIA and IIIB disease predominated, accounting for 112 cases (54.7%) and 53 cases (37.9%), respectively, while 18 patients (7.4%) were diagnosed with stage IIIC disease.
Table 1
| Characteristic | Data |
|---|---|
| Age, years | 60 [33–78] |
| Sex | |
| Female | 74 (30.5) |
| Male | 169 (69.5) |
| Smoking | |
| Yes | 125 (51.4) |
| No | 118 (48.6) |
| ECOG PS | |
| 0 | 97 (39.9) |
| 1 | 146 (60.1) |
| Histology | |
| Adenocarcinoma | 180 (74.1) |
| Squamous cell carcinoma | 55 (22.6) |
| Other | 8 (3.3) |
| Method for evaluating lymph node metastasis | |
| EBUS-TBNA | 158 (65.0) |
| Mediastinoscopy | 85 (35.0) |
| Clinical T classification | |
| T1 | 106 (43.6) |
| T2 | 88 (36.2) |
| T3 | 33 (13.6) |
| T4 | 14 (5.8) |
| Tx | 2 (0.8) |
| Clinical N classification | |
| N2 | 175 (72.0) |
| N3 | 68 (28.0) |
| Involved lymph node station | |
| Single | 31 (12.8) |
| Multiple | 212 (87.2) |
| Clinical TNM stage | |
| IIIA | 133 (54.7) |
| IIIB | 92 (37.9) |
| IIIC | 18 (7.4) |
| Tumor location | |
| Right upper lobe | 38 (15.6) |
| Right middle lobe | 43 (17.7) |
| Right lower lobe | 91 (37.4) |
| Left upper lobe | 20 (8.2) |
| Left lower lobe | 51 (21.0) |
| Neoadjuvant therapy | |
| Chemotherapy | 122 (50.2) |
| IO-chemo | 75 (30.9) |
| Targeted therapy | 46 (18.9) |
Data are presented as median [range] or n (%). ECOG PS, Eastern Cooperative Oncology Group performance status; EBUS-TBNA, endobronchial ultrasound-guided transbronchial needle aspiration; TNM, tumor-node-metastasis; IO-chemo, immuno-chemotherapy.
Neoadjuvant treatment strategies and downstream managements
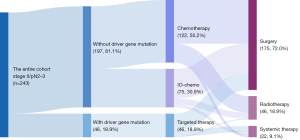
The distribution of treatment modalities and subsequent therapeutics in the study population is illustrated in Figure 2. All of the 46 patients with driver gene mutations [EGFR: 32 cases; ALK: 8 cases; ROS proto-oncogene 1 (ROS1): 2 cases; B-Raf proto-oncogene, serine/threonine kinase V600E mutation (BRAF V600E): 1 case; Others: 3 cases] received targeted therapy with or without chemotherapy (hereafter referred to as the targeted therapy group), including 22 patients treated with targeted therapy alone and 24 patients who received targeted therapy combined with chemotherapy. Among the 197 patients without driver gene mutations, 122 received chemotherapy alone (chemotherapy group), while 75 received immunotherapy in combination with chemotherapy (IO-chemo group). Among patients receiving IO-chemo with available PD-L1 testing (n=34), 8 had tumor proportion score (TPS) ≤1%, 16 had TPS >1–50%, and 10 had TPS >50%. Following neoadjuvant treatments, 175 patients (72.0%) underwent surgical resection, 46 patients (18.9%) transitioned to radiotherapy, and 22 continued systemic therapy. The proportion of patients proceeding to surgery did not differ significantly among the three neoadjuvant groups, with the IO-chemo group having the most surgical patients (77.3%), compared with 69.6% in the targeted therapy group and 69.7% in the chemotherapy group. In the chemotherapy group, 26.2% of patients received radiotherapy after neoadjuvant therapy as a local intervention instead of surgery, whereas this proportion was 16.0% in the IO-chemo group. In the targeted therapy group, 26.1% of patients continued systemic therapy, markedly higher than the chemotherapy group (4.1%) and the IO-chemo group (6.7%).
Survival analyses
The EFS and OS curves for the entire cohort are shown in Figure 3. The median EFS was 28.7 months [95% confidence interval (CI): 26.2–37.4], and the median OS was 67.2 months (95% CI: 57.4–93.6). Survival outcomes were then compared according to the different neoadjuvant treatment modality. Patients in the chemotherapy group exhibited inferior prognosis compared with those in the targeted therapy or IO-chemo groups, and the survival outcomes between the targeted therapy and IO-chemo groups were similar (Figure S1). We further analyzed survival differences based on subsequent treatment modality following neoadjuvant therapy. The median EFS was 24.0 months (95% CI: 15.2–35.8) in the radiotherapy group and 30.1 months (95% CI: 26.6–39.9) in the surgery group (Figure S2A). The median OS was 44.5 months (95% CI: 37.2–62.3) and 84.5 months (95% CI: 57.4–NA) for these two local treatments, respectively (Figure S2B).

Prognosis analyses
Based on univariate Cox analysis for EFS, clinical T classification was identified as a potential risk factor, whereas histology of adenocarcinoma, targeted therapy, and IO-chemo served as protective factors (Table S1). After adjustment in multivariate Cox analysis, targeted therapy (HR =0.46, 95% CI: 0.26–0.83, P=0.009) and IO-chemo (HR =0.54, 95% CI: 0.34–0.85, P=0.008) remained independent protective factors for EFS (Table 2).
Table 2
| Characteristic | EFS | OS | |||||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | P | HR | 95% CI | P | ||
| Age, years | 1.03 | 1.00–1.06 | 0.07 | ||||
| Sex | |||||||
| Female | 1 | ||||||
| Male | 2.27 | 1.04–4.95 | 0.04 | ||||
| Smoking | |||||||
| No | 1 | ||||||
| Yes | 0.98 | 0.52–1.84 | 0.94 | ||||
| Histology | |||||||
| Squamous cell carcinoma | 1 | ||||||
| Adenocarcinoma | 0.88 | 0.58–1.34 | 0.56 | ||||
| Other | 0.78 | 0.23–2.62 | 0.68 | ||||
| Method for evaluating lymph node metastasis | |||||||
| EBUS-TBNA | 1 | ||||||
| Mediastinoscopy | 0.57 | 0.34–0.95 | 0.03 | ||||
| Clinical T classification | |||||||
| T1 | 1 | ||||||
| T2 | 1.24 | 0.81–1.90 | 0.32 | ||||
| T3 | 1.39 | 0.81–2.40 | 0.23 | ||||
| T4 | 2.87 | 1.45–5.69 | 0.002 | ||||
| Tx | 3.16 | 0.74–13.49 | 0.12 | ||||
| Neoadjuvant therapy | |||||||
| Chemotherapy | 1 | 1 | |||||
| IO-chemo | 0.54 | 0.34–0.85 | 0.008 | 0.43 | 0.21–0.89 | 0.02 | |
| Targeted therapy | 0.46 | 0.26–0.83 | 0.009 | 0.47 | 0.18–1.21 | 0.12 | |
| Objective response of neoadjuvant therapy | |||||||
| PR | 1 | ||||||
| SD | 1.39 | 0.84–2.30 | 0.21 | ||||
| PD | 4.31 | 1.45–12.80 | 0.009 | ||||
CI, confidence interval; EBUS-TBNA, endobronchial ultrasound-guided transbronchial needle aspiration; ECOG PS, Eastern Cooperative Oncology Group performance status; EFS, event-free survival; HR, hazard ratio; IO-chemo, immuno-chemotherapy; OS, overall survival; PD, progressive disease; PR, partial response; SD, stable disease; T, tumor.
For OS, univariate Cox analysis indicated that age, sex, smoking history, method for evaluating lymph node metastasis, neoadjuvant treatment regimens, and treatment responses were significantly associated with survival outcomes (Table S1). In multivariate analysis, mediastinoscopy lymph node staging (HR =0.57, 95% CI: 0.34–0.95, P=0.03) and IO-chemo (HR =0.43, 95% CI: 0.21–0.89, P=0.02) were confirmed as independent protective factors for OS, whereas male sex (HR =2.27, 95% CI: 1.04–4.95, P=0.04) and disease progression after neoadjuvant therapy (HR =4.31, 95% CI: 1.45–12.80, P=0.02) were identified as independent risk factors (Table 2).
Subgroup analyses of resected patients
A total of 175 patients ultimately underwent surgical resection. The postoperative pathological responses of these patients are summarized in Table S2. The proportion of patients who achieved pathological downstaging was 79.3% in the IO-chemo group and 81.3% in the targeted therapy group, both higher than that in the chemotherapy group. We further stratified the targeted therapy group and found that the downstaging rate was 69.2% (9/13) among patients who received targeted therapy alone, whereas it reached 89.5% (17/19) in those who received targeted therapy combined with chemotherapy. Further stratification by primary tumor and lymph node status showed that for primary tumor downstaging, the highest rate was observed in the targeted group (78.1%) and the lowest in the chemotherapy group (58.8%), although the difference is not statistically significant among groups. For regional lymph node downstaging, the chemotherapy group still had the lowest proportion (60.0%), which was significantly lower than that observed in the targeted therapy group (P=0.02) and the IO-chemo group (P=0.04). On pathological assessment (Figure S3), the MPR rate was 48.3% in the IO-chemo group and 28.1% in the targeted therapy group, both superior to that in the chemotherapy group (10.6%). A subgroup analysis was performed within the targeted therapy group, and we observed that the MPR rate was 23.1% (3/13) among patients receiving targeted therapy alone, whereas it was 26.3% (5/19) in those treated with combined targeted therapy and chemotherapy. Regarding pCR, the pCR rate in the IO-chemo group was 34.5%, which was significantly higher than that in the chemotherapy group (1.2%, P<0.001) and the targeted therapy group (9.4%, P=0.02). Similarly, we have performed a subgroup analysis within the targeted therapy cohort and found that the pCR rate was 7.7% (1/13) among patients receiving targeted therapy alone, whereas it was 10.5% (2/19) in those treated with combined targeted therapy and chemotherapy.
Subgroup analyses of pN2 patients
A total of 175 patients were confirmed to have pN2 disease, including 57 who received neoadjuvant IO-chemo, 28 who received neoadjuvant targeted therapy, and 90 who received neoadjuvant chemotherapy. Following neoadjuvant treatment, 149 patients underwent surgical resection and 26 received radiotherapy. The 5-year EFS and OS rates for the overall cohort were 28.7% and 67.2%, respectively (Figure S4). Survival analysis showed that patients who underwent surgery after neoadjuvant therapy achieved better EFS and OS compared with those who received radiotherapy, although the differences were not statistically significant (median EFS: surgery vs. radiotherapy =30.0 vs. 23.6 months, P=0.08, Figure S5A; median OS: surgery vs. radiotherapy =70.3 vs. 41.9 months, P=0.15, Figure S5B). In addition, survival was significantly improved among patients who received maintenance therapy after surgery or radiotherapy compared with those who did not (median EFS: yes vs. no = 37.4 vs. 26.6 months, P=0.04, Figure S6A; median OS: yes vs. no = NA vs. 64.1 months, P=0.04, Figure S6B). Multivariate Cox regression analysis identified advanced stage and progressive disease after neoadjuvant therapy as independent risk factors for EFS, whereas neoadjuvant IO-chemo was confirmed as an independent favorable factor for OS (Table S3).
Discussion
Our study provides a comprehensive real-world analysis of MTDs and subsequent outcomes in patients with stage III (N2–3) NSCLC. We demonstrate that neoadjuvant therapy, particularly IO-chemo and targeted therapy, significantly contribute to downstaging, improved pathological responses, and enhanced survival outcomes. Prognosis is promising for patients who underwent surgery after effective neoadjuvant therapy These findings highlight the evolving landscape of stage III NSCLC management and underscore the importance of integrating novel therapies into MDT pathways.
Most contemporary clinical trials in the neoadjuvant setting have predominantly enrolled patients with stage IB–IIIA disease (8-10), whereas evidence for patients with stage III (N2–3) remains limited. Our study specifically focuses on this challenging population, using data from a high-volume cancer center, which reflects real-world practice patterns more accurately. Our findings may therefore provide valuable insights into the clinical course and treatment outcomes of patients with advanced stage III disease. In addition, our study may also emphasize the importance of real-world analyses in complementing clinical trial data and guiding individualized decision-making for these complex cases.
Although all patients included in our study had pN2/3 disease, the median OS still reached 67.2 months. We believe several factors may contribute to these favorable outcomes. First, the application of more effective systemic therapies, including immunotherapy and targeted therapy, results in higher MPR and pCR rates compared with traditional chemotherapy alone (8,9), thereby enabling a greater proportion of patients to undergo curative-intent surgery (with a median OS exceeding 72 months among resected cases in our study). Second, these novel agents may have also improved control of micro-metastatic disease, translating into better long-term survival (15). Third, advances in minimally invasive surgical techniques have facilitated faster recovery and reduced perioperative morbidity and non-lung cancer-related mortality, further improving patient outcomes (16-18). These encouraging survival results also raise important considerations. Current guidelines (5,19) recommend concurrent chemoradiotherapy followed by consolidation immunotherapy or targeted therapy as the standard treatment for patients with N2–3 disease. For those with single-station, non-bulky N2, upfront neoadjuvant systemic therapy may be considered, with surgery or radiotherapy if disease remains stable. Historically, in the era of neoadjuvant chemotherapy, surgery following induction chemotherapy for locally advanced NSCLC conferred only a modest survival benefit, with an absolute improvement of approximately 5% in 5-year OS (from about 40% to 45%) compared with surgery alone (20). In contrast, in the current era of neoadjuvant IO + chemo, long-term survival outcomes have improved substantially, with recent trials reporting 5-year OS rates of approximately 65–68.4% (21,22). In our study, the median OS of the resected N2–3 cases exceeded 5 years. Therefore, our findings suggest that patients with N2–3 disease may not be categorically excluded from surgery, as they may still derive meaningful survival benefit, particularly those who respond well to novel neoadjuvant therapy. However, treatment selection bias cannot be excluded due to the retrospective nature of this study. We believe that further research, a head-to-head comparison of surgery versus radiotherapy after induction therapy, is warranted to draw a definitive conclusion.
Notably, we find that neoadjuvant IO-chemo achieved the highest rates of MPR and pCR, followed by targeted therapy, with chemotherapy being the least effective. These findings are consistent with recent phase II–III trials (8-10). For example, in the NeoADAURA study (9), the MPR rate was 26% in the osimertinib plus chemotherapy arm and 25% with osimertinib monotherapy, compared with 28.1% in our cohort; likewise, pCR rates were 4% and 9% in the NeoADAURA combination and monotherapy arms, respectively, versus 9.4% in our study. The consistency of these findings suggest that neoadjuvant targeted therapy in NSCLC can achieve reproducible clinical benefits beyond those of neoadjuvant chemotherapy, with good generalizability in real-world practice. However, our multivariate Cox analysis reveals no significant difference in OS between patients receiving neoadjuvant IO-chemo versus targeted therapy. This raises the important question of whether MPR is a valid surrogate endpoint for OS across different therapeutic modalities (23,24). In particular, the definition of MPR as ≤10% viable tumor cells may not be fully appropriate for targeted therapy, suggesting the need to re-examine pathological response thresholds in future studies (23).
In addition, our data show that patients receiving maintenance therapy after surgery or radiotherapy had superior survival compared with those without maintenance, consistent with current guideline recommendations (5). This underscores the importance of MDT discussions after local interventions (surgery or radiotherapy) to ensure that optimal maintenance strategies are implemented. Another noteworthy observation is that, over a 15-year period, the proportion of cN2–3 patients who underwent mediastinoscopy or EBUS-TBNA prior to treatment was relatively low. This finding highlights the need for stricter adherence to guideline-recommended staging procedures in routine clinical practice to improve treatment stratification and outcomes.
Our study has several limitations. First, its retrospective, single-center design may introduce selection bias, and the relatively small sample size, along with limitations in data quality, may further restrict the generalizability of our findings. Second, patients with stage III NSCLC who did not undergo pathological confirmation of pN2–3 disease were excluded, which may reduce the representativeness of the study population. This reflects real-world practice where many patients proceed directly to treatment based on clinical staging without invasive nodal assessment. However, the use of EBUS or mediastinoscopy for accurate nodal evaluation ensured reliable pathological staging among the included patients, which likely reduced staging misclassification and enhanced the robustness of our findings. Third, this study does not systematically assess treatment-related adverse events or toxicity profiles, which are critical for evaluating the safety and tolerability of different neoadjuvant strategies. At last, the long study period might introduce potential temporal bias, as patients in the chemotherapy group were more likely to be treated in earlier years, whereas patients receiving IO-chemo or targeted therapy were treated in later periods. Advances in surgical techniques and supportive care over time may also have contributed to improved outcomes in more recent cohorts. These factors should be considered when interpreting our findings. Future multicenter retrospective studies and well-designed prospective clinical trials are warranted to validate the efficacy of MDT-based approaches in stage III NSCLC.
Conclusions
In summary, this real-world study of stage III (N2–3) NSCLC demonstrates that novel neoadjuvant strategies, particularly immunotherapy and targeted therapy, resulted in superior pathological responses and long-term survival compared with chemotherapy. Importantly, even patients with N3 disease could derive survival benefit from surgery when combined with effective neoadjuvant systemic therapy. These findings emphasize the value of multidisciplinary management, highlight the role of maintenance therapy in consolidating outcomes, and provide evidence to inform future studies aimed at optimizing treatment strategies for this challenging population.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the TREND reporting checklist. Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-aw-1230/rc
Data Sharing Statement: Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-aw-1230/dss
Peer Review File: Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-aw-1230/prf
Funding: This work was supported in part by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-aw-1230/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the Ethics Committee of Sun Yat-sen University Cancer Center (No. B2025-452-01) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Siegel RL, Kratzer TB, Giaquinto AN, et al. Cancer statistics, 2025. CA Cancer J Clin 2025;75:10-45. [Crossref] [PubMed]
- Han B, Zheng R, Zeng H, et al. Cancer incidence and mortality in China, 2022. J Natl Cancer Cent 2024;4:47-53. [Crossref] [PubMed]
- Narra LR, Kumar R, Deek MP, et al. Updates in Management of Unresectable Stage III Non Small Cell Lung Cancer: A Radiation Oncology Perspective. Cancers (Basel) 2024;16:4233. [Crossref] [PubMed]
- Rami-Porta R, Nishimura KK, Giroux DJ, et al. The International Association for the Study of Lung Cancer Lung Cancer Staging Project: Proposals for Revision of the TNM Stage Groups in the Forthcoming (Ninth) Edition of the TNM Classification for Lung Cancer. J Thorac Oncol 2024;19:1007-27. [Crossref] [PubMed]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: NonSmall Cell Lung Cancer. Version 3.2025. Published January 14, 2025. Accessed April 19, 2025. Available online: https://www.nccn.org/guidelines/guidelinesdetail?category=1&id=1450
- Chinese Society of Clinical Oncology. (2025). CSCO NSCLC Guidelines 2025. Available online: https://www.csco.org.cn
- Garassino MC, Torri V. Neoadjuvant or Perioperative Approach in Lung Cancer. N Engl J Med 2024;390:1816-8.
- Forde PM, Spicer JD, Provencio M, et al. Overall Survival with Neoadjuvant Nivolumab plus Chemotherapy in Lung Cancer. N Engl J Med 2025;393:741-52. [Crossref] [PubMed]
- He J, Tsuboi M, Weder W, et al. Neoadjuvant Osimertinib for Resectable EGFR-Mutated Non-Small Cell Lung Cancer. J Clin Oncol 2025;43:2875-87. [Crossref] [PubMed]
- Spicer JD, Garassino MC, Wakelee H, et al. Neoadjuvant pembrolizumab plus chemotherapy followed by adjuvant pembrolizumab compared with neoadjuvant chemotherapy alone in patients with early-stage non-small-cell lung cancer (KEYNOTE-671): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2024;404:1240-52. [Crossref] [PubMed]
- World Medical Association Declaration of Helsinki. ethical principles for medical research involving human subjects. JAMA 2013;310:2191-4. [Crossref] [PubMed]
- Goldstraw P, Chansky K, Crowley J, et al. The IASLC Lung Cancer Staging Project: Proposals for Revision of the TNM Stage Groupings in the Forthcoming (Eighth) Edition of the TNM Classification for Lung Cancer. J Thorac Oncol 2016;11:39-51. [Crossref] [PubMed]
- Travis WD, Dacic S, Wistuba I, et al. IASLC Multidisciplinary Recommendations for Pathologic Assessment of Lung Cancer Resection Specimens After Neoadjuvant Therapy. J Thorac Oncol 2020;15:709-40. [Crossref] [PubMed]
- Mantel N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 1966;50:163-70.
- Phillips WJ, Jackson A, Kidane B, et al. Immunotherapy for Early-Stage Non-Small Cell Lung Cancer: A Practical Guide of Current Controversies. Clin Lung Cancer 2025;26:179-90. [Crossref] [PubMed]
- Ricciardi R, Seshadri-Kreaden U, Yankovsky A, et al. The COMPARE Study: Comparing Perioperative Outcomes of Oncologic Minimally Invasive Laparoscopic, da Vinci Robotic, and Open Procedures: A Systematic Review and Meta-analysis of the Evidence. Ann Surg 2025;281:748-63. [Crossref] [PubMed]
- Yang MZ, Tan ZH, Li JB, et al. Comparison of Short-Term Outcomes Between Robot-Assisted and Video-Assisted Segmentectomy for Small Pulmonary Nodules: A Propensity Score-Matching Study. Ann Surg Oncol 2023;30:2757-64. [Crossref] [PubMed]
- Sun TY, Xie CL, Tan Z, et al. Short-term outcomes of robotic lobectomy versus video-assisted lobectomy in patients with pulmonary neoplasms. Thorac Cancer 2023;14:1512-9. [Crossref] [PubMed]
- Zer A, Ahn MJ, Barlesi F, et al. Early and locally advanced non-small-cell lung cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol 2025;36:1245-62. [Crossref] [PubMed]
- Preoperative chemotherapy for non-small-cell lung cancer: a systematic review and meta-analysis of individual participant data. Lancet 2014;383:1561-71. [Crossref] [PubMed]
- Wakelee H, Liberman M, Kato T, et al. Perioperative Pembrolizumab for Early-Stage Non-Small-Cell Lung Cancer. N Engl J Med 2023;389:491-503. [Crossref] [PubMed]
- Forde PM, Spicer J, Lu S, et al. Neoadjuvant Nivolumab plus Chemotherapy in Resectable Lung Cancer. N Engl J Med 2022;386:1973-85. [Crossref] [PubMed]
- Dai C, Yang X, Yang D, et al. Association Between Pathologic Response and Survival Following Neoadjuvant Targeted Therapy in Epidermal Growth Factor Receptor-Mutant Lung Adenocarcinoma. Eur J Cardiothorac Surg 2025;67:ezaf269. [Crossref] [PubMed]
- Cai JS, Li S, Yan SM, et al. Is major pathologic response sufficient to predict survival in resectable nonsmall-cell lung cancer patients receiving neoadjuvant chemotherapy? Thorac Cancer 2021;12:1336-46. [Crossref] [PubMed]