
Comparison of lobectomy performed through Toumai® surgical robot and da Vinci surgical robot in early-stage non-small cell lung cancer: a retrospective study of early perioperative results
Highlight box
Key findings
• The Toumai® surgical robot system has a comparable short-term outcome compared with the da Vinci robotic surgical system for patients with early-stage non-small cell lung cancer (NSCLC).
What is known and what is new?
• The da Vinci robotic surgical system has shown its safety and efficiency in routine operations such as lobectomy.
• This is the first retrospective study of early perioperative results for the evaluation of the efficacy of Toumai® robotic system in NSCLC.
What is the implication, and what should change now?
• Toumai® surgical robot will be an optional method for robotic-assisted thoracoscopic surgery, and more researches need to be conducted in the future.
Introduction
Nowadays, minimally invasive thoracic approaches such as video-assisted thoracic surgery (VATS) and robotic-assisted thoracic surgery (RATS) are recognized to be safe, with best short-term outcomes and preserved long-term outcomes for lung resections for early-stage non-small cell lung cancer (NSCLC). RATS represents the evolution of VATS by proposing technical solutions that go beyond the limits of VATS. Today in many studies those two approaches are still discussed (1-3), highlighting the question of the optimal minimally invasive surgical approach which is a challenging subject.
The da Vinci robotic surgical system (Intuitive Surgical, Sunnyvale, CA, USA) is the commercially available robotic surgical system, and other new robots including modular technology have been recently commercialized in Europe and in some other countries (4). RATS approach has been developed and spread fast. However, the platform’s high cost and lack of accessibility are currently holding back its development (5,6).
The Toumai® surgical robot system for minimally invasive operations was developed by Shanghai Minimally Invasive Medical Robot Co., Ltd. (Shanghai, China). In prior tests, expert robotic surgeons have been able to smoothly complete routine operations such as lobectomy with the Toumai® surgical robot system (7).
Shanghai Chest Hospital introduced the da Vinci S surgical robot in 2009 and completed the first robot-assisted lobectomy in mainland of China (8). Since then, using the da Vinci Si and Xi surgical robots, more than 5,000 robot-assisted operations have been completed for thoracic diseases in the Shanghai Lung Cancer Center in Shanghai Chest Hospital.
In this study, we enrolled 19 cases with early-stage NSCLC and aimed to provide definitive evidence on the surgical and oncological safety of the Toumai® surgery robot compared with the mature da Vinci robot for clinical early-stage NSCLC. We present this article in accordance with the STROBE reporting checklist (available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-23-603/rc).
Methods
Study design
The study is a single-center, retrospective comparative cohort study. We included consecutive patients aged from 18 to 80 years with NSCLC undergoing RATS lobectomy in our center, the Shanghai Chest Hospital, between November 2021 and December 2021.
Inclusion/exclusion criteria
A total of 19 patients who underwent RATS lobectomy during this period were enrolled. The assessment includes previous medical history, physical examination, pulmonary function test, and computed tomography (CT) of the chest. All cases were discussed in multidisciplinary staff. In accordance with the 8th edition of TNM staging system of The International Association for the Study of Lung Cancer, all patients were evaluated with a clinical stage IA to IIA by enhanced thoracic CT and positron emission tomography/CT (PET/CT), and endobronchial ultrasound-guided transbronchial puncture was conducted if necessary.
The exclusion criteria included: tumor invasion into neighboring organs, including hilum; history of thoracic surgery or high-dose radiation; pregnant or lactating female patients; history of other malignancies; extensive pleural adhesion or pleural metastasis found during surgery; pathological diagnosis other than NSCLC.
All these 19 patients met the above criteria. They were willing to undergo surgery with the help of any robotic platform and signed informed consent forms after preoperative introduction. Depending on the type of surgical robot, the patients were divided into Toumai® group (n=9) and Da Vinci group (n=10). Although it was not a prospective study, robot-assisted lobectomy was firstly considered suitable for these 19 patients and then a random draw was used to determine the type of robot. Informed consent was taken from all the patients. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Institutional Review Board of Shanghai Chest Hospital (filing number: LS2169).
Surgical procedures
A radical lobectomy associated with a complete lymph node (LN) dissection was the standard surgical intervention according to the national comprehensive cancer network guidelines for NSCLC (9). In both groups, we followed the definition of the Cancer and Leukemia Group B 39802, including anatomic lobectomy, visualization only by thoracoscope and non-rib-spreading technique (10).
A chief surgeon and an expert assistant who were well trained in animal tests for Toumai® surgical robot participated in the whole trial. They also have collaborated on more than 300 da Vinci robot-assisted surgeries in recent 2 years, and the chief surgeon has the experience of more than 1,500 da Vinci robot-assisted surgeries. A da Vinci Si or Xi surgical robot and a Toumai® surgical robot were used, and surgical procedures were similar in both robotic platforms. We chose the 4-arm lobectomy technique and the number of ports was defined in the trial (11,12) (Figure 1). The camera port was placed in the 7th intercostal space (ICS) of the midaxillary line. Two working ports were placed in the 6th ICS of the anterior axillary line and the 8th ICS of the posterior axillary line, through which the electrocautery hook and the robotic forceps were inserted. A 30-mm auxiliary port was placed in the 3rd or 4th ICS of the anterior axillary line, which helped the assistant to use the aspirator and the thoracoscopic stapler (Johnson & Johnson, New Brunswick, NJ, USA). For every patient, we dissected appropriate N1 and N2 LN stations.
Outcomes
We focused our analysis on short-term perioperative outcomes, including conversion rate, operation time (docking time, console time and closure time), blood loss during operation, thoracic drainage, complication within 4 weeks of surgery, postoperative length of stay (LOS), and the number of LNs and LN stations harvested. Clavien-Dindo scale for the classification of postoperative complications was used to further define complications (13). Within the 5 years after the lobectomy, patients were reviewed in our center in 6-month intervals. The 5-year overall survival (OS) and disease-free survival (DFS) will be released in the future.
Statistical analysis
Due to the small sample size, the comparison of outcomes between groups was performed by the Mann-Whitney U test and the Fisher exact test. Continuous data was summarized as median and interquartile range (IQR). Categorical data was summarized as numbers. A 2-sided P value of less than 0.05 was considered statistically significant. Statistical analysis was performed with the software SPSS 27.0 (IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics
A total of 19 patients were recruited, all of whom were eligible for analyses (Toumai® group, n=9; da Vinci group, n=10). There was no significant difference in both groups concerning age, sex, body mass index (BMI), smoking history, chronic disease, and pulmonary function. In tumor-related aspects, no statistical difference was identified in tumor size, tumor location, clinical stage, histology and pathological stage (Table 1).
Table 1
| Variables | Toumai® (n=9) | da Vinci (n=10) | P |
|---|---|---|---|
| Age (year) | 62 [44.50–74.50] | 61.5 [55.75–66.75] | 0.842 |
| Sex, n | >0.99 | ||
| Male | 5 | 5 | |
| Female | 4 | 5 | |
| BMI (kg/m2) | 22.58 [19.94–26.69] | 23.31 [21.17–26.92] | 0.905 |
| FEV1 (% of predicted) | 91.4 [86.5–97.6] | 90.05 [88.05–104.13] | 0.968 |
| DLCO (% of predicted) | 94 [84.85–111.75] | 108.15 [96.28–111.65] | 0.356 |
| Smoking history | 0.141 | ||
| Ever | 4 | 1 | |
| Never | 5 | 9 | |
| Hypertension | 0.370 | ||
| Yes | 5 | 3 | |
| No | 4 | 7 | |
| Diabetes | >0.99 | ||
| Yes | 1 | 1 | |
| No | 8 | 9 | |
| Tumor location | 0.510 | ||
| Right upper lobe | 4 | 2 | |
| Right middle lobe | 2 | 1 | |
| Right lower lobe | 1 | 3 | |
| Left lower lobe | 2 | 4 | |
| Tumor size (mm) | 16 [11.5–21.5] | 20 [12.75–28.5] | 0.549 |
| Clinical stage | >0.99 | ||
| IA | 9 | 9 | |
| IB | 0 | 0 | |
| IIA | 0 | 1 | |
| Histology | >0.99 | ||
| MIA | 1 | 1 | |
| Invasive adenocarcinoma | 8 | 8 | |
| Typical carcinoid | 0 | 1 | |
| Pathological stage | 0.285 | ||
| IA | 8 | 5 | |
| IB | 1 | 3 | |
| IIA | 0 | 1 | |
| IIB | 0 | 0 | |
| IIIA | 0 | 1 |
Continuous data is presented as median [interquartile range], and categorical data is presented as numbers. BMI, body mass index; FEV1, forced expiratory volume in 1 second; DLCO, carbon monoxide diffusing capacity; MIA, minimally invasive adenocarcinoma.
Perioperative outcomes
The perioperative outcomes are reported in Table 2. No procedure was converted to thoracotomy, and all of these patients underwent a R0 resection safely. In the Toumai® group and the da Vinci group, no significant differences were observed concerning operation time: docking time [10 minutes (IQR, 6.5–16.5 minutes) vs. 9 minutes (IQR, 7.75–11 minutes), P=0.497], console time [66 minutes (IQR, 52–98 minutes) vs. 55.5 minutes (IQR, 51–79.25 minutes), P=0.211], closure time [23 minutes (IQR, 18.5–26 minutes) vs. 21 minutes (IQR, 15.5–22.5 minutes), P=0.356]. The two groups also had comparable blood loss (P=0.211), postoperative drainage characteristics (P=0.079), chest tube duration (P=0.356) and postoperative LOS (P=0.905). Prolonged air leak (grade I) in two patients from both groups was the only complication reported.
Table 2
| Variables | Toumai® (n=9) | da Vinci (n=10) | P value |
|---|---|---|---|
| Total operation time (min) | 95 [86.5–136.5] | 86 [81–102] | 0.178 |
| Docking time (min) | 10 [6.5–16.5] | 9 [7.75–11] | 0.497 |
| Console time (min) | 66 [52–98] | 55.5 [51–79.25] | 0.211 |
| Closure time (min) | 23 [18.5–26] | 21 [15.5–22.5] | 0.356 |
| Blood loss | 0.211 | ||
| ≤100 mL | 7 | 10 | |
| >100–200 mL | 2 | 0 | |
| Conversion | 0 | 0 | – |
| R0 resection | 9 | 10 | – |
| Clavien-Dindo complications | >0.99 | ||
| None | 7 | 8 | |
| I | 2 | 2 | |
| II to V | 0 | 0 | |
| Postoperative LOS (day) | 4 [3.5–7.5] | 4 [4–4.5] | 0.905 |
| Postoperative drainage (mL) | 605 [355–785] | 310 [216.25–400] | 0.079 |
| Chest tube duration (day) | 4 [3–5.5] | 3.5 [3–4] | 0.356 |
| Number of sampled LN stations | 7 [4.5–8] | 6.5 [5.75–8] | 0.842 |
| Number of sampled LNs | 8 [6–9.5] | 8 [5.75–10.5] | 0.842 |
| Nodal up-staging | |||
| cN0→pN1 | 0 | 0 | – |
| cN0→pN2 | 0 | 1 | – |
Continuous data is presented as median [interquartile range], and categorical data is presented as numbers. LOS, length of stay; LN, lymph node.
LNs assessment
LN dissection was done in the same manner in both groups, and no differences in the number of LNs [8 (IQR, 6–9.5) vs. 8 (IQR, 5.75–10.5), P=0.842] and the harvested LN stations [7 (IQR, 4.5–8) vs. 6.5 (IQR, 5.75–8), P=0.842] was reported (Table 2). Among the 19 cases, only one patient in the da Vinci group was up-stagged from cN0 to pN2.
Discussion
The da Vinci robotic surgery system has developed rapidly in recent years and has been applied in the field of thoracic surgery in China for more than 14 years (8). Compared with VATS approach, robot-assisted surgery allows to overcome VATS technical limitations with its high-definition, stable 3-dimensional (3D) imaging endoscope system, highly flexible mechanical wrists graspers, double-handed energy graspers, stable robotic staplers and a supplementary arm for exposure. All those advantages allow the surgeons to mimic an open procedure by a closed-chest approach (14). However, concerning short-term and long-term oncological outcomes, the comparison between RATS and VATS is a complex issue and both approaches allow the surgeons to perform a safe and oncological lung resection (15-18).
In recent years, the Chinese government has issued robot related industrial policies to vigorously support the development of robot enterprises. The Toumai® surgical robot is a Chinese surgical robot independently developed by Shanghai Minimally Invasive Medical Robot Co., Ltd. Because the da Vinci robotic platform is very expensive in most hospitals, many patients can not bear the cost without private medical insurance (6,19). As a home-made surgical robotic platform, Toumai® surgical robot will show better economical advantages because part of the surgical cost will be reimbursed by Chinese medical insurance in the future.
In addition, 5G is expected to be used for remote control, allowing a second expert surgeon to participate in the operation from distant locations, which will provide convenience to patients in remote regions and also reduce their total cost (20). If its safety and efficiency can be further verified through a series of follow-up clinical trials in the future, this new robotic platform will become an alternative choice for patients in need of remote medical treatment.
In the aspect of the operation time, thanks to years of experience in da Vinci robotic surgery, the surgeon and his assistant were rapidly proficient in the use of Toumai® surgical robot after a short wet-lab training on animals (20 procedures). The 9 cases included in this study preliminarily showed the safety and stability of the Toumai® robot. The median operation time of lobectomy plus systematic LN dissection was 95 minutes (IQR, 86.5–136.5 minutes), and no case was converted to thoracotomy. Liang et al. (21) performed an analysis of 14 studies including 3,239 patients undergoing da Vinci robotic surgery. The meta-analysis showed that the average operation time of RATS anatomical pulmonary resection was 176.6 minutes, and the intraoperative conversion rate was as high as 10.3%. Our center summarized 1,000 cases of da Vinci robot-assisted lung surgery (5). The average time of robot surgery was 90.3 minutes, and the intraoperative conversion rate was 0.9%. Compared with da Vinci robot surgery, regardless of the data within China and internationally, these 9 cases in the Toumai® group meet the requirements in operation time and conversion rate. There were no statistical difference concerning perioperative outcomes, as the median operation time, including docking time (P=0.497), console time (P=0.211) and closure time (P=0.356). As reported in the literature concerning the learning curve (22-25), as the surgeons become more familiar with the use of the Toumai® Chinese robot, we are looking forward to further shorten the operation time and enhanced postoperative short-term outcomes.
Because of the lack of certified training program, robotic surgery needs a long learning curve in the initial surgical practice (22,23). The learning curve can be evaluated using various factors, such as operating time, the emergency’s conversion rate, the completeness of the resection, and the postoperative complications. The learning curve is debated, but the threshold of about 20 operations is often mentioned in the literature (24,25). Except for the differences in appearance of the two robotic platforms, the operation performed with the Toumai® robot is performed in the same way as using the da Vinci robotic platform from the perspective of the surgeon, his assistant and the scrub nurses (Figures 2,3). The Toumai® wrist surgical instruments are highly flexible with seven degrees of freedom, double-handed energy graspers and the robotic stapler is also available. The independently developed DFVision® 3D endoscope system (Shanghai Minimally Invasive Medical Robot Co., Ltd.) provides a 3D, stable and real surgical field vision. The intuitive master-slave teleoperation is sensitive and easy to use, so as to simplify the surgical operation and shorten the operation time. Because the development of the Toumai® robot aims to follow surgeons’ habits in previous da Vinci robotic-assisted surgeries, the settings and the working field are familiar to experienced surgeons. These characteristics will reduce the learning curve on this new robotic platform. Representative procedural steps of right upper lobectomy and right middle lobectomy performed by Toumai® robot are presented in Videos 1,2.

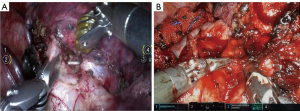
The patients in the Toumai® group experienced a longer chest tube duration [4 days (IQR, 3–5.5 days) vs. 3.5 days (IQR, 3–4 days), P=0.356] and had more postoperative drainage [605 mL (IQR, 355–785 mL) vs. 310 mL (IQR, 216.25–400 mL), P=0.079] compared with those in another group, but not statistically significant in this study. The energy devices used by both robots were double-handed energy graspers, such as electric hooks and Maryland forceps, and the assistant can use high intensity focused ultrasound and argon plasma coagulation to assist in hemostasis. No serious intraoperative bleeding or postoperative complications occurred in either groups, so we think that these energy devices are safe and will be more familiar to surgeons in the future. In addition, as clinicians were cautious to this new robotic platform, the chest tube duration and LOS may have been extended. Whether the amount of blood loss and drainage has a statistical difference needs to be discussed in further studies with large sample sizes.
LN invasion plays an important prognostic role. The adequate LN dissection is mandatory to perform an oncological resection. Some reports have shown that a complete LN dissection can be performed more easily and accurately by RATS compared to VATS (26-28). Moreover, using the da Vinci robotic platform intraoperative bleeding are reduced compared to VATS allowing an enhanced recovery (29,30). In the nine cases of Toumai® robot surgery, the median number of resected LNs is 8 (IQR, 6–9.5) and harvested LNs stations is 7 (IQR, 4.5–8) and these results are no different from the da Vinci surgery group.
Nevertheless, there are several limitations in this study. This is a retrospective study of small number of cases in a single institution, and more clinical outcomes should be obtained to assess the advantages and disadvantages of Toumai® surgical robot. In addition, we focus only on perioperative short-term outcomes in this article, and long-term outcomes such as 5-year OS and DFS will be released in the future.
Conclusions
As a domestically developed surgical robot in China, Toumai® surgical robot seems to have similar short-term and oncological outcomes in this small cohort compared with da Vinci robotic surgery system for early-stage NSCLC.
Application of the Toumai® surgical robot in lobectomy was preliminarily shown to be safe and efficient, and a longer follow-up to conclude about the OS and DFS is necessary. Further studies with a bigger cohort will confirm the preliminary clinical real-life data.
The Toumai® surgical robot has the potential to be an optional method for the new generation of minimally invasive robotic thoracic surgery in the future.
Acknowledgments
The authors appreciate the great support from Dr. Fernando Vannucci [National Cancer Institute (INCA), Brazil] in improving the quality of this paper.
Funding: This work was supported by
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-23-603/rc
Data Sharing Statement: Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-23-603/dss
Peer Review File: Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-23-603/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-23-603/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Informed consent was taken from all the patients. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Institutional Review Board of Shanghai Chest Hospital (filing number: LS2169).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Flores RM, Alam N. Video-assisted thoracic surgery lobectomy (VATS), open thoracotomy, and the robot for lung cancer. Ann Thorac Surg 2008;85:S710-5. [Crossref] [PubMed]
- Pan H, Tian Y, Wang H, et al. Perioperative and Oncological Outcomes of Robotic-Assisted, Video-Assisted Thoracoscopic and Open Lobectomy for Patients with N1-Metastatic Non-Small Cell Lung Cancer: A Propensity Score-Matched Study. Cancers (Basel) 2022;14:5249. [Crossref] [PubMed]
- Oh DS, Reddy RM, Gorrepati ML, et al. Robotic-Assisted, Video-Assisted Thoracoscopic and Open Lobectomy: Propensity-Matched Analysis of Recent Premier Data. Ann Thorac Surg 2017;104:1733-40. [Crossref] [PubMed]
- Aresu G, Dunning J, Routledge T, et al. Preclinical evaluation of Versius, an innovative device for use in robot-assisted thoracic surgery. Eur J Cardiothorac Surg 2022;62:ezac178. [Crossref] [PubMed]
- Li C, Huang J, Li J, et al. Robotic lung resection for malignant and benign lesions: experience with 1000 patients. Chinese Journal of Clinical Thoracic and Cardiovascular Surgery 2019;26:42-7.
- Heiden BT, Mitchell JD, Rome E, et al. Cost-Effectiveness Analysis of Robotic-assisted Lobectomy for Non-Small Cell Lung Cancer. Ann Thorac Surg 2022;114:265-72. [Crossref] [PubMed]
- Huang J, Tian Y, Lu P, et al. Two cases of right upper lobe resection assisted by Toumai® endoscopic surgery robot. Chinese Journal of Clinical Thoracic and Cardiovascular Surgery 2022;29:519-23.
- Lin H, Huang J, Tan Q, et al. Robot-assisted left lower lobectomy with report of 2 cases. Chinese Journal of Laparoscopic Surgery 2012;5:288-93.
- Ettinger DS, Wood DE, Aisner DL, et al. Non-Small Cell Lung Cancer, Version 5.2017, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2017;15:504-35. [Crossref] [PubMed]
- Swanson SJ, Herndon JE 2nd, D'Amico TA, et al. Video-assisted thoracic surgery lobectomy: report of CALGB 39802--a prospective, multi-institution feasibility study. J Clin Oncol 2007;25:4993-7. [Crossref] [PubMed]
- Veronesi G, Galetta D, Maisonneuve P, et al. Four-arm robotic lobectomy for the treatment of early-stage lung cancer. J Thorac Cardiovasc Surg 2010;140:19-25. [Crossref] [PubMed]
- Cerfolio RJ, Bryant AS, Skylizard L, et al. Initial consecutive experience of completely portal robotic pulmonary resection with 4 arms. J Thorac Cardiovasc Surg 2011;142:740-6. [Crossref] [PubMed]
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 2004;240:205-13. [Crossref] [PubMed]
- Montagne F, Baste JM. Should we keep on doing robotic surgery to treat lung cancer in 2020? Ann Transl Med 2020;8:775. [Crossref] [PubMed]
- Farivar AS, Cerfolio RJ, Vallières E, et al. Comparing robotic lung resection with thoracotomy and video-assisted thoracoscopic surgery cases entered into the Society of Thoracic Surgeons database. Innovations (Phila) 2014;9:10-5. [Crossref] [PubMed]
- Zhang J, Feng Q, Huang Y, et al. Updated Evaluation of Robotic- and Video-Assisted Thoracoscopic Lobectomy or Segmentectomy for Lung Cancer: A Systematic Review and Meta-Analysis. Front Oncol 2022;12:853530. [Crossref] [PubMed]
- Baig MZ, Razi SS, Agyabeng-Dadzie K, et al. Robotic-assisted thoracoscopic surgery demonstrates a lower rate of conversion to thoracotomy than video-assisted thoracoscopic surgery for complex lobectomies. Eur J Cardiothorac Surg 2022;62:ezac281. [Crossref] [PubMed]
- Montagne F, Chaari Z, Bottet B, et al. Long-Term Survival Following Minimally Invasive Lung Cancer Surgery: Comparing Robotic-Assisted and Video-Assisted Surgery. Cancers (Basel) 2022;14:2611. [Crossref] [PubMed]
- Le Gac C, Gondé H, Gillibert A, et al. Medico-economic impact of robot-assisted lung segmentectomy: what is the cost of the learning curve? Interact Cardiovasc Thorac Surg 2020;30:255-62. [Crossref] [PubMed]
- Tian Y, Huang J, Li J, et al. Animal experimental study of 5G remote robot-assisted thoracoscopic lobectomy. Chinese Journal of Clinical Thoracic and Cardiovascular Surgery 2023;30:1112-5.
- Liang H, Liang W, Zhao L, et al. Robotic Versus Video-assisted Lobectomy/Segmentectomy for Lung Cancer: A Meta-analysis. Ann Surg 2018;268:254-9. [Crossref] [PubMed]
- Augustin F, Bodner J, Wykypiel H, et al. Initial experience with robotic lung lobectomy: report of two different approaches. Surg Endosc 2011;25:108-13. [Crossref] [PubMed]
- Ninan M, Dylewski MR. Total port-access robot-assisted pulmonary lobectomy without utility thoracotomy. Eur J Cardiothorac Surg 2010;38:231-2. [Crossref] [PubMed]
- Gharagozloo F, Margolis M, Tempesta B, et al. Robot-assisted lobectomy for early-stage lung cancer: report of 100 consecutive cases. Ann Thorac Surg 2009;88:380-4. [Crossref] [PubMed]
- Park BJ, Flores RM, Rusch VW. Robotic assistance for video-assisted thoracic surgical lobectomy: technique and initial results. J Thorac Cardiovasc Surg 2006;131:54-9. [Crossref] [PubMed]
- Aiolfi A, Nosotti M, Micheletto G, et al. Pulmonary lobectomy for cancer: Systematic review and network meta-analysis comparing open, video-assisted thoracic surgery, and robotic approach. Surgery 2021;169:436-46. [Crossref] [PubMed]
- Yang HX, Woo KM, Sima CS, et al. Long-term Survival Based on the Surgical Approach to Lobectomy For Clinical Stage I Nonsmall Cell Lung Cancer: Comparison of Robotic, Video-assisted Thoracic Surgery, and Thoracotomy Lobectomy. Ann Surg 2017;265:431-7. [Crossref] [PubMed]
- Jin R, Zheng Y, Yuan Y, et al. Robotic-assisted Versus Video-assisted Thoracoscopic Lobectomy: Short-term Results of a Randomized Clinical Trial (RVlob Trial). Ann Surg 2022;275:295-302. [Crossref] [PubMed]
- Mahieu J, Rinieri P, Bubenheim M, et al. Robot-Assisted Thoracoscopic Surgery versus Video-Assisted Thoracoscopic Surgery for Lung Lobectomy: Can a Robotic Approach Improve Short-Term Outcomes and Operative Safety? Thorac Cardiovasc Surg 2016;64:354-62. [Crossref] [PubMed]
- Jang HJ, Lee HS, Park SY, et al. Comparison of the early robot-assisted lobectomy experience to video-assisted thoracic surgery lobectomy for lung cancer: a single-institution case series matching study. Innovations (Phila) 2011;6:305-10. [Crossref] [PubMed]



