Treatment interruption during neoadjuvant immuno-chemotherapy for non-small-cell lung cancer: a systematic review and meta-analysis of randomized controlled trials
Highlight box
Key findings
• Neoadjuvant immuno-chemotherapy, compared with neoadjuvant chemotherapy alone, did not increase the risk of neoadjuvant treatment discontinuation or surgery delay, and was associated with a lower risk of surgery cancellation, particularly in cases of cancellation caused by progression of disease.
What is known and what is new?
• Neoadjuvant therapy is an important treatment strategy for non-small-cell lung cancer (NSCLC). Treatment interruption during neoadjuvant therapy is a recognized concern impacting NSCLC treatment plans and potentially outcomes.
• This work provides a systematic comparison of the differential risk of pre-operative treatment interruption between neoadjuvant immuno-chemotherapy and other neoadjuvant strategies for NSCLC, an area where current evidence remains unclear.
What is the implication, and what should change now?
• These findings reveal the additional advantages of neoadjuvant immuno-chemotherapy in the treatment of early-stage NSCLC from the perspective of pre-operative treatment interruption.
Introduction
Lung cancer is the most commonly diagnosed neoplasm worldwide, which is also the leading cause of cancer death (1,2). In recent years, neoadjuvant therapy (NAT), the systemic therapy for patients with resectable cancer before surgery, has become a common management strategy for early-stage resectable lung cancer (3). Previous neoadjuvant therapies primarily relied on chemotherapy. However, neoadjuvant chemotherapy has shown unsatisfactory results in terms of downstaging rate, pathological complete response (pCR) rate, major pathological response (MPR) rate, disease-free survival (DFS) and overall survival (OS) (4,5).
Immune checkpoint inhibitors (ICIs), as a new systemic treatment approach, have shown great potential in the treatment of lung cancer (6). Studies have reported that neoadjuvant immunotherapy demonstrates acceptable efficacy in terms of safety, R0 resection rate, pCR rate, and MPR rate (7-9). Neoadjuvant immunotherapy combined chemotherapy is currently the most promising neoadjuvant treatment strategy. Several studies had demonstrated that neoadjuvant immuno-chemotherapy offered better event free survival (EFS), pCR rate, and MPR rate compared to chemotherapy alone (10,11). Based on these findings, neoadjuvant immune-chemotherapy has been approved by the Food and Drug Administration as the standard NAT strategy for early-stage resectable non-small-cell lung cancer (NSCLC). However, it is important to note that despite the overall benefits of neoadjuvant immuno-chemotherapy in resectable NSCLC, a significant proportion of patients in studies experienced pre-operative treatment interruption (10,12-21).
Pre-operative treatment interruption mainly included NAT discontinuation, surgery delay, and surgery cancellation. The primary causes of treatment interruption are typically adverse events (AEs), progressive disease (PD) and other objective reasons (22-24). NAT discontinuation is associated with a higher risk of R1/2 resections, increased ypT/N stage, and poor treatment response (23). Although there has been no consensus on the optimal time window for NSCLC resection after NAT, a meta-analysis including seven types of cancer showed that a surgical delay of more than 4 weeks was associated with increased mortality (25). In addition, surgery cancellation is associated with more complications, higher treatment costs, and poorer survival outcomes (22,26,27). Pre-operative treatment interruption has significant impacts on both patient prognosis and the healthcare system. However, the differences in the risk of pre-operative treatment interruption among NAT strategies remain unclear.
This systematic review and meta-analysis assessed the published randomized controlled trials (RCTs) of neoadjuvant immuno-chemotherapy, intended to provide timely evidence on the incidence of pre-operative treatment interruption, including NAT discontinuation, surgery delay and surgery cancellation between neoadjuvant immuno-chemotherapy and chemotherapy alone in NSCLC. In the subgroup analysis, we also explored the differences in pre-operative treatment interruption risks caused by specific reasons.
Research protocol of present study has been registered with PROSPERO (CRD42024570066). We present this article in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) reporting checklist (28) (available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-919/rc).
Methods
Search strategy and study selection
We searched MEDLINE (PubMed), Embase and the Cochrane Library on July 3, 2024 with keywords “non-small-cell lung cancer”, “neoadjuvant therapy” and “immunotherapy”. Complete search strategy was available in Tables S1-S3. To assess the incidence of pre-operative treatment interruption, only phase 2 and 3 RCTs were included, as patients with comparable baseline between groups were intervened and assessed at the same time, which was less likely to be biased by confounding factors. Studies reporting at least one of NAT discontinuation and surgery cancellation and surgery delay was included. Studies reporting small cell lung cancer or inoperable NSCLC were excluded. For articles reporting the same clinical trial, we selected the one with latest and widest result. As we only included prospective controlled studies, the Cochrane risk-of-bias tool was used for article assessment (29).
Data extraction and endpoints definition
Information of research name, first author name, year of publication, PMID, registration number, study design, tumor histology, name and target of ICIs, treatment regimen, treatment design, number of patients receiving NAT in each group, number of NAT discontinuation, surgery cancellation and surgery delay was retrieved. To rule out patients not undergoing treatment, we only selected numbers of each arm within as-treated population. NAT discontinuation was defined as subjects not completing planned NAT. Surgery cancellation was defined as subjects not completing tumor resection surgery. Surgery delay was defined as subjects not receiving tumor resection surgery within designed time window. The specific reasons of pre-operative treatment interruption were also extracted. Literature search, bias assessment, data review and extraction were conducted by two authors (Yi Liu and Z.W.) independently, conflicting results were evaluated by a third author (S.M.).
Statistical analysis
We used a paired meta-analysis to investigate the difference in the risk of pre-operative treatment interruption between neoadjuvant ICI plus chemotherapy and chemotherapy alone. Next, we further conducted a network meta-analysis (NMA) to compare the impact of four different NAT regimens [ICI plus chemotherapy, chemotherapy alone, ICI alone, ICI plus stereotactic body radiotherapy (SBRT)] on the risk of pre-operative treatment interruption. Given limited number of published RCTs, different ICIs were integrated. To further analysis the difference on specific reason of pre-operative treatment interruption, we divided the numbers as AE-related and PD-related pre-operative treatment interruption. Another subgroup of combining AE and PD-related pre-operative treatment interruption was also constructed, as they were two major concerns of NAT.
The Mantel-Haenszel method was used for paired meta-analysis, with Z test applied for hypothesis-testing (30). The Cochrane Q test was performed to assess the heterogeneity within included studies. Heterogeneity was acknowledged when I2 over 50% and P value less than 0.1 was simultaneously observed. Random effect model was used if heterogeneity existed (31). The formula of I2 was:
To avoid potential confounding bias, the age, gender, histology type, smoking status and study population of patients were extracted for meta-regression if available. For publication bias, Egger test was conducted, with Funnel plot used for visualization.
To compare the difference of pre-operative treatment interruption among these neoadjuvant therapies, the Bayesian NMA was performed, and Markov chain Monte Carlo Method was used for computing 95% confidence interval (CI) (32). Bayesian method was able to reduce estimation bias and overconfident conclusions and more appropriate when dealing with sparse data (33,34). The I2 and Cochrane Q statistics were calculated for assessing heterogeneity. Deviance information criterion (DIC) was used to appraise the fitting degree (35), which was taken into consideration whether applying random effect or fixed effect model (33,36). To conduct NMA, four independent chains were run with iterations of 100,000. To improve the sampling efficiency of subsequent iterations, the iterations was set as 5,000 in the adopting stage. The initial 1,000 iterations were discarded to make sure the initial value does not affect results. To visualize NMA results, the cumulative ranking plot was used and surface under the cumulative ranking (SUCRA) was calculated to rank different neoadjuvant therapies from the least risk of interruption to the most likely. The convergence of model was evaluated by the shape of posterior distribution and Brooks-Gelman-Rubin method. The value of potential scale reduction factor (PSRF) approaching one indicated satisfactory model convergence (37). To adjust the confounders of age, gender, histology type, smoking status and study population. Bayesian network meta-regression was also performed to estimate transitivity of indirect comparison (38). As head-to-head RCT was not available, the inconsistency test was not carried out (38,39).
R 4.2.3 (https://www.R-project.org/) was used. Package “meta” version 7.0.0 (40), “metafor” version 4.6-0 (41), “BUGSnet” version 1.1.2 (42) and “gemtc” version 1.0-2 (43) were used to perform meta-analyses and draw forest plot. Risk ratio (RR) and 95% CI was used to quantify the difference of pre-operative treatment interruption (44). The absolute risk difference (ARD) was used to compare the absolute difference in the occurrence of pre-operative treatment interruption between the two groups, with results presented as the number of events per 1,000 patients. Beta and standard error were used in measuring the result of meta-regression. All statistical tests were two-sided with the P value less than 0.05 indicating significant if not mentioned elsewhere.
Results
Literature search results
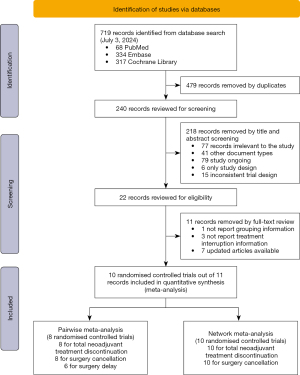
The results of literature search were displayed in Figure 1. A total of 719 literatures were enrolled as first. After removing irrelevant, duplicated and outdated literatures, ten RCTs out of eleven literatures were finally included for analysis, including four phase 2 trials (10,12-15) and six phase 3 trials (16-21). The basic information of each trial was listed in Table 1, including 3,582 NSCLC patients receiving NAT within 22 arms. For the type of ICIs, 549 patients were treated with nivolumab, while 460 for durvalumab, 396 for Pembrolizumab, 226 for tislelizumab, 202 for toripalimab, 43 for camrelizumab, and 22 for ipilimumab. In contrast, there were 1,706 patients receiving neoadjuvant CT, and 30 patients receiving ICIs plus SBRT. For the NAT discontinuation, one RCT reported the number of ICI/placebo and chemotherapy separately (17), considering the potential overlap of discontinuation of both therapies, the larger number was used.
Table 1
| No. | Author [year] | PMID | Trial name | Study registration number | Trial type | Histology type | ICI type | ICI dose regimen | Treatment design | Sample size | Available outcomes |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Forde et al. [2022] | 35403841 | CheckMate 816 | NCT02998528 | Phase 3, open label | Resectable stage IB (≥4 cm) to IIIA NSCLC without sensitizing EGFR mutations or ALK translocations | Nivolumab | 360 mg, every 3 weeks for three cycles | ICI plus CT vs. CT | 176/176 | Neoadjuvant therapy discontinuation; surgery cancellation; surgery delay |
| 2 | Cascone et al. [2021] | 36928818 | NEOSTAR | NCT03158129 | Phase 2, open label | Resectable stage IB (≥4 cm) to IIIA NSCLC | Nivolumab/ipilimumab | Nivolumab 360 mg IV every 3 weeks; ipilimumab 1 mg/kg on day 1 of first cycle only | One ICI plus CT vs. two ICIs plus CT | 22/22 | Neoadjuvant therapy discontinuation; surgery cancellation |
| 3 | Cascone et al. [2021] | 33603241 | NEOSTAR | NCT03158129 | Phase 2, open label | Resectable stage IB (≥4 cm) to IIIA NSCLC | Nivolumab/ipilimumab | Nivolumab 360 mg IV every 3 weeks; ipilimumab 1 mg/kg on day 1 of first cycle only | One ICI vs. two ICIs | 22/22 | Neoadjuvant therapy discontinuation; surgery cancellation |
| 4 | Lei et al. [2023] | 37535377 | TD-FOREKNOW | NCT04338620 | Phase 2, open label | Resectable stage IIIA or IIIB (stage IIIB, T3N2 only) NSCLC | Camrelizumab | 200 mg on day 1 of each 3-week cycle for 3 cycles | ICI plus CT vs. CT | 43/45 | Neoadjuvant therapy discontinuation; surgery cancellation; surgery delay |
| 5 | Heymach et al. [2023] | 37870974 | AEGEAN | NCT03800134 | Phase 3, double-blind | Resectable stage II to IIIB (N2 node stage) NSCLC | Durvalumab | 1,500 mg every 3 weeks for four cycles | ICI plus CT vs. placebo plus CT | 400/399 | Neoadjuvant therapy discontinuation; surgery cancellation; surgery delay |
| 6 | Altorki et al. [2023] | 34015311 | – | NCT02904954 | Phase 2, open label | Resectable stage IA to IIIA NSCLC | Durvalumab | 1.12 g, 3 weeks for two cycles | ICI vs. ICI plus SBRT | 30/30 | Neoadjuvant therapy discontinuation; surgery cancellation, surgery delay |
| 7 | Provencio et al. [2023] | 37379158 | NADIM II | NCT03838159 | Phase 2, open label | Resectable stage IIIA or IIIB NSCLC | Nivolumab | 360 mg, 21 days (with a window of ±3 days) for three cycles | ICI plus CT vs. CT | 57/29 | Neoadjuvant therapy discontinuation; surgery cancellation; surgery delay |
| 8 | Wakelee et al. [2023] | 37272513 | KEYNOTE-671 | NCT03425643 | Phase 3, double-blind | Resectable pathologically confirmed stage II, IIIA, or IIIB (N2 node stage) NSCLC | Pembrolizumab | 200 mg, every 3 weeks for four cycles | ICI plus CT vs. placebo plus CT | 396/399 | Neoadjuvant therapy discontinuation; surgery cancellation |
| 9 | Cascone et al. [2024] | 38749033 | CheckMate 77T | NCT04025879 | Phase 3, double-blind | Resectable stage IIA to IIIB (N2 node stage) NSCLC | Nivolumab | 360 mg, every 3 weeks for four cycles | ICI plus CT vs. placebo plus CT | 228/230 | Neoadjuvant therapy discontinuation; surgery cancellation; surgery delay |
| 10 | Yue et al. [2024] | conference | RATIONALE-315 | NCT04379635 | Phase 3, double-blind | Resectable stage II–IIIA NSCLC, no known EGFR mutations or ALK gene translocations | Tislelizumab | 200 mg, 3–4 cycles | ICI plus CT vs. placebo plus CT | 226/226 | Neoadjuvant therapy discontinuation; surgery cancellation; surgery delay |
| 11 | Lu et al. [2024] | 38227033 | Neotorch | NCT04158440 | Phase 3, double-blind | Resectable stage III NSCLC | Toripalimab | 240 mg, every 3 weeks for 3 cycles | ICI plus CT vs. placebo plus CT | 202/202 | Neoadjuvant therapy discontinuation; surgery cancellation |
The table presents the basic information of 11 included studies (10 RCTs). CT, chemotherapy; ICI, immune checkpoint inhibitor; NSCLC, non-small-cell lung cancer; RCT, randomized controlled trial; SBRT, stereotactic body radiotherapy.
As a result, 484 times of NAT discontinuation was recorded. While 711 times of surgery cancellation and 313 times of surgery delay were reported. In specific, surgery delay was reported in six RCTs (13,15-17,19,20). Normally, surgery was planned within 6 weeks after the completion of NAT (13,16,17,19,20), while in one study, surgery was conducted within 4 weeks of the last administration in the last cycle (15). The Cochrane risk of bias assessment results indicated low risk of bias in all RCTs included in this study (Figure S1).
Risk of pre-operative treatment interruption between immuno-chemotherapy vs. chemotherapy
To compare the risk of pre-operative treatment interruption between immuno-chemotherapy vs. chemotherapy, we excluded articles without chemotherapy arm, which left eight RCTs, CheckMate 816 study (16), TD-FOREKNOW study (45), AEGEAN study (17), NADIM II study (15), KEYNOTE-671 study (18), CheckMate 77T study (19), RATIONALE-315 study (20) and Neotorch study (21). Bias of these articles is provided in Figure S2.
As in Figure 2, compared to neoadjuvant chemotherapy, neoadjuvant immuno-chemotherapy exhibited no significant difference in overall NAT discontinuation (RR, 1.04; 95% CI: 0.78–1.40; I2=42%). Interestingly, a relatively higher risk of AE-related discontinuation (RR, 1.32; 95% CI: 1.00–1.73; I2=4%) and a lower risk of PD-related (RR, 0.53; 95% CI: 0.30–0.96; I2=0%) discontinuation were observed in the immuno-chemotherapy group. When both AE-related and PD-related reasons were considered together, the risk of NAT discontinuation showed no significant difference (RR, 1.07; 95% CI: 0.84–1.35; I2=30%).

In the aspect of surgery interruption, while neoadjuvant immuno-chemotherapy increased the risk of AE-related surgery cancellation (RR, 1.81; 95% CI: 1.18–2.76; I2=0%), the overall (RR, 0.79; 95% CI: 0.70–0.90; I2=26%), PD-related (RR, 0.48; 95% CI: 0.32–0.73; I2=51%), and AE plus PD-related (RR, 0.71; 95% CI: 0.58–0.86; I2=27%) surgery cancellation risk were lower in contrast to neoadjuvant chemotherapy (Figure 3). The ARD analyses revealed that for every 1,000 patients receiving NAT, administrated with immuno-chemotherapy instead of chemotherapy alone could reduce the event of surgery cancellation in approximately 38.5 patients. Detailed results are presented in Table S4. For surgery delay, due to limited number of RCT, only overall and AE-related event were compared, however, there were no significant difference (Figure 4).


The meta-regression results showed that age, gender, histology type, smoking status, and study population had no significant impact on the effect size (Table S5). In a nutshell, neoadjuvant immuno-chemotherapy did not demonstrate higher risk of pre-operative treatment interruption than neoadjuvant chemotherapy, instead, it could reduce the risk of PD-related NAT discontinuation and surgery cancellation.
Risk of pre-operative treatment interruption among different neoadjuvant therapies
To compare the pre-operative treatment interruption risk of different NAT, we grouped them as immuno-chemotherapy, immunotherapy, chemotherapy and immunotherapy plus SBRT. We conduced NMA comparing NAT discontinuation and surgery cancellation among these four groups. And no statistic of PD-related NAT discontinuation in immunotherapy and immunotherapy plus SBRT was available for network analysis (Figure 5A).

To conduct Bayesian NMA, the DIC was calculated for both the fixed effect and random effect model (Table S6), and model with smaller DIC value was chosen for analysis. The convergence of the model was diagnosed and visualized in Figure S3. The network diagrams of NMA were provided in Figure 5A,5B, followed by superiority ranking of the four NAT regimens (Figure 5C-5J). Immunotherapy alone seemed to have lowest risk of NAT discontinuation compared with other three NAT but not significant (Figure S4). On the other hand, chemotherapy tend to have the lowest risk of AE-related surgery cancellation, while immuno-chemotherapy might be better in overall, PD-related and AE plus PD-related surgery cancellation (Figure S5). In summary, the results of Bayesian NMA demonstrated that the risk of pre-operative treatment interruption of neoadjuvant immuno-chemotherapy was not significantly higher than the other three treatment regimens. After adjusting for potential confounding factors such as age, gender, histology type, smoking status, and study population, none of the covariates showed a statistically significant association with the primary outcome (P>0.05). The transitivity assumption was validated in regression analysis, indicating that the indirect comparisons between different treatment strategies exhibited good transitivity and consistency. The detailed results of network meta-regression analyses were available in Table S7.
Besides, we summarized the common causes of pre-operative treatment interruption of different regiments and their proportions in Table S8, and further provided a breakdown of the specific reasons for AE-related pre-operative treatment interruptions caused by different neoadjuvant strategies (Table S9).
Discussion
This systematic review and meta-analysis of RCTs focused on pre-operative treatment interruption in NSCLC, an endpoint of substantial clinical relevance that has often been underestimated. Treatment interruption provides a practical composite endpoint, integrating both efficacy and toxicity into a single measure of surgical feasibility. Delayed or canceled surgery due to PD or treatment-related AEs directly reflects deterioration in prognosis and can therefore serve as a surrogate marker of therapeutic balance. Our results showed no difference in overall NAT discontinuation and surgery delay between neoadjuvant immuno-chemotherapy and chemotherapy alone. Immuno-chemotherapy increased the risk of AE-related NAT discontinuation and surgery cancellation, but reduced the risk of PD-related NAT discontinuation and overall surgery cancellation, compared with chemotherapy. In the NMA, immuno-chemotherapy did not significantly increase the risk of NAT discontinuation compared to other treatment strategies and potentially exhibited the lowest risk of overall, PD-related, and AE plus PD-related surgery cancellations.
Neoadjuvant chemotherapy is the standard of care of resectable NSCLC, with its advantages in OS compared with surgery alone (46). Current major targets of immunotherapy including programmed cell death protein 1 (PD-1) (nivolumab, camrelizumab, pembrolizumab, tislelizumab, toripalimab), programmed death-ligand 1 (PD-L1) (durvalumab), and cytotoxic T lymphocyte-associated molecule 4 (CTLA-4) (ipilimumab) (47). Recent advance in neoadjuvant immuno-chemotherapy has been validated in several RCTs, on its survival (15-17), and pathologic response (15,17,18,45) benefits over neoadjuvant chemotherapy, indicating huge potential. The same conclusion on improving prognosis was also drawn in meta-analysis (48).
By inhibition of immune checkpoints, the immune system was activated against tumor, on the other hand, undesired effect may also arise. The most common AE of ICIs was observed within gastrointestinal tract, endocrine glands, skin, and liver (49). Although previous research reported increased risk of AE of neoadjuvant immuno-chemotherapy over chemotherapy alone for NSCLC, the difference in grade 3 to 4, grade 5, or overall TRAEs was not significant (48). Another meta-analysis of NSCLC reported neoadjuvant immuno-chemotherapy tended to have higher incidence of TRAE comparing chemotherapy alone but not significant. And no difference in the incidence of TRAE of neoadjuvant immuno-chemotherapy versus immunotherapy (50). Despite NAT has been found safe and effective for operable NSCLC, disease progression was another risk, as tumor cells were keeping proliferating due to treatment resistance or other reasons. The total rate of PD precluding surgery was believed to be lower for neoadjuvant immuno-chemotherapy versus chemotherapy (48). On the other hand, the pathological endpoint of NAT was also documented. MPR was defined as less than 10% residual viable tumor cells in both the primary tumor and sampled lymph nodes, and pCR was defined as 0% residual viable tumor cells, both of them were reliable predictors of NSCLC survival (51). As mentioned above, meta-analysis confirmed neoadjuvant immuno-chemotherapy with higher MPR and pCR than chemotherapy or immunotherapy alone (48,50). Taken together, this duality reflects the balance of immuno-chemotherapy: while it reduced PD-related interruption, it also introduced a modest increase in AE-related discontinuation.
NAT discontinuation was an unpleasant event with patients failed to complete administrated treatment before surgery. Commonly known reasons of NAT discontinuation included AE, PD, consent withdraw/refusal of patient, patient death and so on (12,13,17,18). Both incomplete NAT and the reason causing it could result in removal from research group in clinical trials and disturb the schedule of surgery, and finally lead to worse prognosis. No difference was found in the risk of patients receiving all cycles of NAT between immuno-chemotherapy and chemotherapy (48), which was consistent with our results. Nevertheless, immuno-chemotherapy slightly increased the risk of AE-related NAT discontinuation over chemotherapy. It could be suspected that combination of two agents could lead to more serious AE, however, one meta-analysis suggested the toxicity rates of neoadjuvant immuno-chemotherapy and chemotherapy was similar (52). Importantly, when interpreting these findings, the absolute incidence of NAT discontinuation and surgery cancellation was relatively low across trials; therefore, although relative risks suggest a higher AE-related stop with immuno-chemotherapy, the actual number of patients affected may be small in clinical practice.
Subsequently, patients were receiving tumor resection after NAT, but surgery could be cancelled or delayed due to variable reasons. For neoadjuvant immuno-chemotherapy of NSCLC, one of the major reasons of surgery cancellation was PD, while AE was the most common underlying cause of surgery delay and the least reported cause of surgery cancellation (53). In our study of neoadjuvant immuno-chemotherapy, the delay rate of different therapy was not different, which was not conflict with previous research (50). So far, there was no consensus on the best time window for surgical resection after NAT, which could possibly be determined by the type of NAT, healthy status of patients and predicted stage of disease, while most RCTs we included have chosen the time within 6 weeks after finishing NAT. It is safe to conclude that based on the current workflow of NSCLC NAT, immuno-chemotherapy did not increase the risk of surgery delay over chemotherapy.
It has been reported that compared with neoadjuvant chemotherapy, immuno-chemotherapy reduced the risk of patients not undergoing surgery (48). Notably, there were differences in the definition of not undergoing surgery and surgery cancellation, as we included patients who underwent surgery but failed to conduct tumor resection. In KEYNOTE-671 study, five patients in immuno-chemotherapy arm were recorded as unresectable due to have metastatic disease at the time of surgery and thirteen in the chemotherapy arm (18). We believe this definition of surgery cancellation was appropriate as their tumor mass was not physically removed. We also found this reduced risk of immuno-chemotherapy than chemotherapy, in addition, this reduction was probably contributed by reduced risk of PD-related surgery cancellation.
SBRT has been used as complement for early-stage NSCLC patients not suitable for surgery with no difference in survival from two prospective cohorts (54). The combination of SBRT and neoadjuvant immunotherapy has been applied in an attempt to suppress the growth of local tumor while cooperate with activated systemic immune response (55). In NMA, immunotherapy plus SBRT was seemed to be associated with higher risk of AE or PD-related pre-operative treatment interruption, which could probably be biased as only one RCT of neoadjuvant SBRT was included (4). Further study with comparable baseline, treatment design and time window of surgery could provide more accurate information on this matter.
Limitations of this study included several aspects. First, a potentially important moderator of pre-operative treatment interruption and surgical timing is baseline disease stage (for example stage IB versus stage IIIA). Clinically, more advanced stages are associated with larger tumor burden and higher risk of early progression, which could increase the probability of PD-related discontinuation or surgery cancellation. Conversely, patients with more advanced but technically resectable disease are often managed in high-volume centers with intensive multidisciplinary monitoring, which may mitigate the risk of treatment interruption or delay (56). However, the lack of stage-stratified reporting for pre-operative interruption and surgical timing is an important limitation of existing randomized evidence. Second, perioperative outcomes, including postoperative complication rates, conversion from minimally invasive to open surgery, and the need for extended resections, were not consistently reported across randomized trials, precluding a quantitative synthesis of these endpoints. Third, biomarker information such as PD-L1 status, which could plausibly influence NAT tolerance and surgical feasibility, was not reported in a stratified manner for treatment interruption or surgery outcomes in the included RCTs. Fourth, although we restricted our analysis to prospective RCTs given their high level of evidence, the relatively small number of eligible trials and treatment arms (especially for immunotherapy monotherapy or immunotherapy plus SBRT) may have introduced imprecision. Additionally, heterogeneity in inclusion criteria, type and dose of ICIs, NAT design, and surgery time window across studies may also contribute to baseline differences and variability in treatment interruption outcomes. Finally, due to the limited literature base, inconsistency testing within the NMA could not be performed.
Taken together, these limitations highlight the urgent need for future RCTs and large-scale real-world datasets to provide more granular and standardized reporting—specifically stage-stratified, PD-L1-stratified data, as well as detailed perioperative outcomes including complications, conversion to thoracotomy, and extent of resection—so that more robust and clinically meaningful subgroup analyses of pre-operative treatment interruption can be achieved.
Conclusions
Our results suggested that neoadjuvant immuno-chemotherapy was associated with a lower overall risk of surgery cancellation, particularly the risk of surgery cancellation due to PD. On the other hand, neoadjuvant immuno-chemotherapy did not increase the overall risk of NAT discontinuation or surgery delay. These findings support the potential advantages of neoadjuvant immuno-chemotherapy over other neoadjuvant treatment strategies in the treatment of early-stage NSCLC, and further confirmation through more comprehensively randomized controlled trials is still needed.
Acknowledgments
We are grateful to Dr. Yunhe Mao for providing statistical expertise.
Footnote
Reporting Checklist: The authors have completed the PRISMA reporting checklist. Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-919/rc
Peer Review File: Available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-919/prf
Funding: This work was supported by
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tlcr.amegroups.com/article/view/10.21037/tlcr-2025-919/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2024;74:229-63. [Crossref] [PubMed]
- Zhang Y, Vaccarella S, Morgan E, et al. Global variations in lung cancer incidence by histological subtype in 2020: a population-based study. Lancet Oncol 2023;24:1206-18. [Crossref] [PubMed]
- Schuler M. Facts and Hopes in Neoadjuvant Immunotherapy Combinations in Resectable Non-Small Cell Lung Cancer. Clin Cancer Res 2025;31:801-7. [Crossref] [PubMed]
- Rossi G, Barcellini L, Tagliamento M, et al. Immunotherapy for resectable NSCLC: neoadjuvant/perioperative followed by surgery over surgery followed by adjuvant. Systematic review and meta-analysis with subgroup analyses. ESMO Open 2025;10:105759. [Crossref] [PubMed]
- Saw SPL, Ong BH, Chua KLM, et al. Revisiting neoadjuvant therapy in non-small-cell lung cancer. Lancet Oncol 2021;22:e501-16. [Crossref] [PubMed]
- Reck M, Remon J, Hellmann MD. First-Line Immunotherapy for Non-Small-Cell Lung Cancer. J Clin Oncol 2022;40:586-97. [Crossref] [PubMed]
- Desai AP, Adashek JJ, Reuss JE, et al. Perioperative Immune Checkpoint Inhibition in Early-Stage Non-Small Cell Lung Cancer: A Review. JAMA Oncol 2023;9:135-42. [Crossref] [PubMed]
- Chaft JE, Oezkan F, Kris MG, et al. Neoadjuvant atezolizumab for resectable non-small cell lung cancer: an open-label, single-arm phase II trial. Nat Med 2022;28:2155-61. [Crossref] [PubMed]
- Forde PM, Chaft JE, Smith KN, et al. Neoadjuvant PD-1 Blockade in Resectable Lung Cancer. N Engl J Med 2018;378:1976-86. [Crossref] [PubMed]
- Cascone T, William WN Jr, Weissferdt A, et al. Neoadjuvant nivolumab or nivolumab plus ipilimumab in operable non-small cell lung cancer: the phase 2 randomized NEOSTAR trial. Nat Med 2021;27:504-14. [Crossref] [PubMed]
- Cascone T, Kar G, Spicer JD, et al. Neoadjuvant Durvalumab Alone or Combined with Novel Immuno-Oncology Agents in Resectable Lung Cancer: The Phase II NeoCOAST Platform Trial. Cancer Discov 2023;13:2394-411. [Crossref] [PubMed]
- Cascone T, Leung CH, Weissferdt A, et al. Neoadjuvant chemotherapy plus nivolumab with or without ipilimumab in operable non-small cell lung cancer: the phase 2 platform NEOSTAR trial. Nat Med 2023;29:593-604. [Crossref] [PubMed]
- Lei J, Zhao J, Gong L, et al. Neoadjuvant Camrelizumab Plus Platinum-Based Chemotherapy vs Chemotherapy Alone for Chinese Patients With Resectable Stage IIIA or IIIB (T3N2) Non-Small Cell Lung Cancer: The TD-FOREKNOW Randomized Clinical Trial. JAMA Oncol 2023;9:1348-55. [Crossref] [PubMed]
- Altorki NK, McGraw TE, Borczuk AC, et al. Neoadjuvant durvalumab with or without stereotactic body radiotherapy in patients with early-stage non-small-cell lung cancer: a single-centre, randomised phase 2 trial. Lancet Oncol 2021;22:824-35. [Crossref] [PubMed]
- Provencio M, Nadal E, González-Larriba JL, et al. Perioperative Nivolumab and Chemotherapy in Stage III Non-Small-Cell Lung Cancer. N Engl J Med 2023;389:504-13. [Crossref] [PubMed]
- Forde PM, Spicer J, Lu S, et al. Neoadjuvant Nivolumab plus Chemotherapy in Resectable Lung Cancer. N Engl J Med 2022;386:1973-85. [Crossref] [PubMed]
- Heymach JV, Harpole D, Mitsudomi T, et al. Perioperative Durvalumab for Resectable Non-Small-Cell Lung Cancer. N Engl J Med 2023;389:1672-84. [Crossref] [PubMed]
- Wakelee H, Liberman M, Kato T, et al. Perioperative Pembrolizumab for Early-Stage Non-Small-Cell Lung Cancer. N Engl J Med 2023;389:491-503. [Crossref] [PubMed]
- Cascone T, Awad MM, Spicer JD, et al. Perioperative Nivolumab in Resectable Lung Cancer. N Engl J Med 2024;390:1756-69. [Crossref] [PubMed]
- Yue D, Tan L, Xu S, et al. Surgical outcomes from RATIONALE-315: Randomized, double-blind, phase III study of perioperative tislelizumab with neoadjuvant chemotherapy in resectable NSCLC. ESMO Open 2024; Conference: European Lung Cancer Congress (ELCC) 2024.
- Lu S, Zhang W, Wu L, et al. Perioperative Toripalimab Plus Chemotherapy for Patients With Resectable Non-Small Cell Lung Cancer: The Neotorch Randomized Clinical Trial. JAMA 2024;331:201-11. [Crossref] [PubMed]
- Wong DJN, Harris SK, Moonesinghe SR, et al. Cancelled operations: a 7-day cohort study of planned adult inpatient surgery in 245 UK National Health Service hospitals. Br J Anaesth 2018;121:730-8. [Crossref] [PubMed]
- Wang J, Wu Z, de Groot EM, et al. Discontinuation of neoadjuvant therapy does not influence postoperative short-term outcomes in elderly patients (≥ 70 years) with resectable gastric cancer: a population-based study from the dutch upper gastrointestinal cancer audit (DUCA) data. Gastric Cancer 2024;27:1114-23. [Crossref] [PubMed]
- Fu SJ, George EL, Maggio PM, et al. The Consequences of Delaying Elective Surgery: Surgical Perspective. Ann Surg 2020;272:e79-80. [Crossref] [PubMed]
- Hanna TP, King WD, Thibodeau S, et al. Mortality due to cancer treatment delay: systematic review and meta-analysis. BMJ 2020;371:m4087. [Crossref] [PubMed]
- Eskander A, Zanchetta C, Coburn N, et al. Cancer surgery cancellation: incidence, outcomes and recovery in a universal health care system. Can J Surg 2022;65:E782-91. [Crossref] [PubMed]
- Nardini M, Chaudhuri N, Lodhia J, et al. Last-minute cancellation of elective lung cancer surgery is associated with poorer survival. Interdiscip Cardiovasc Thorac Surg 2025;40:ivae172. [Crossref] [PubMed]
- Haddaway NR, Page MJ, Pritchard CC, et al. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst Rev 2022;18:e1230. [Crossref] [PubMed]
- Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 2019;366:l4898. [Crossref] [PubMed]
- Kuritz SJ, Landis JR, Koch GG. A general overview of Mantel-Haenszel methods: applications and recent developments. Annu Rev Public Health 1988;9:123-60. [Crossref] [PubMed]
- Deeks J, Higgins J, Altman D. Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins J, Thomas J. editors. Cochrane Handbook for Systematic Reviews of Interventions (6.4). Cochrane Training; 2023.
- Cipriani A, Higgins JP, Geddes JR, et al. Conceptual and technical challenges in network meta-analysis. Ann Intern Med 2013;159:130-7. [Crossref] [PubMed]
- Dias S, Sutton AJ, Ades AE, et al. Evidence synthesis for decision making 2: a generalized linear modeling framework for pairwise and network meta-analysis of randomized controlled trials. Med Decis Making 2013;33:607-17. [Crossref] [PubMed]
- Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 2011;64:163-71. [Crossref] [PubMed]
- Spiegelhalter DJ, Best NG, Carlin BP, et al. Bayesian measures of model complexity and fit. Journal of the Royal Statistical Society 2002;64:583-639.
- Shim SR, Kim SJ, Lee J, et al. Network meta-analysis: application and practice using R software. Epidemiol Health 2019;41:e2019013. [Crossref] [PubMed]
- Gelman A, Rubin DB. Inference from Iterative Simulation Using Multiple Sequences. Statistical Science 1992;7:457-72.
- Efthimiou O, Debray TP, van Valkenhoef G, et al. GetReal in network meta-analysis: a review of the methodology. Res Synth Methods 2016;7:236-63. [Crossref] [PubMed]
- Salanti G, Higgins JP, Ades AE, et al. Evaluation of networks of randomized trials. Stat Methods Med Res 2008;17:279-301. [Crossref] [PubMed]
- Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health 2019;22:153-60. [Crossref] [PubMed]
- Viechtbauer W. Conducting Meta-Analyses in R with the metafor Package. Journal of Statistical Software 2010;36:1-48.
- Béliveau A, Boyne DJ, Slater J, et al. BUGSnet: an R package to facilitate the conduct and reporting of Bayesian network Meta-analyses. BMC Med Res Methodol 2019;19:196. [Crossref] [PubMed]
- van Valkenhoef G, Dias S, Ades AE, et al. Automated generation of node-splitting models for assessment of inconsistency in network meta-analysis. Res Synth Methods 2016;7:80-93. [Crossref] [PubMed]
- Andrade C. Understanding Misunderstandings About the Relative Risk and Odds Ratio. J Clin Psychiatry 2023;84:23f14943.
- Rosner S, Reuss JE, Zahurak M, et al. Five-Year Clinical Outcomes after Neoadjuvant Nivolumab in Resectable Non-Small Cell Lung Cancer. Clin Cancer Res 2023;29:705-10. [Crossref] [PubMed]
- Preoperative chemotherapy for non-small-cell lung cancer: a systematic review and meta-analysis of individual participant data. Lancet 2014;383:1561-71. [Crossref] [PubMed]
- Cheng W, Kang K, Zhao A, et al. Dual blockade immunotherapy targeting PD-1/PD-L1 and CTLA-4 in lung cancer. J Hematol Oncol 2024;17:54. [Crossref] [PubMed]
- Sorin M, Prosty C, Ghaleb L, et al. Neoadjuvant Chemoimmunotherapy for NSCLC: A Systematic Review and Meta-Analysis. JAMA Oncol 2024;10:621-33. [Crossref] [PubMed]
- Boutros C, Tarhini A, Routier E, et al. Safety profiles of anti-CTLA-4 and anti-PD-1 antibodies alone and in combination. Nat Rev Clin Oncol 2016;13:473-86. [Crossref] [PubMed]
- Jiang J, Wang Y, Gao Y, et al. Neoadjuvant immunotherapy or chemoimmunotherapy in non-small cell lung cancer: a systematic review and meta-analysis. Transl Lung Cancer Res 2022;11:277-94. [Crossref] [PubMed]
- Deutsch JS, Cimino-Mathews A, Thompson E, et al. Association between pathologic response and survival after neoadjuvant therapy in lung cancer. Nat Med 2024;30:218-28. [Crossref] [PubMed]
- Wu Y, Verma V, Gay CM, et al. Neoadjuvant immunotherapy for advanced, resectable non-small cell lung cancer: A systematic review and meta-analysis. Cancer 2023;129:1969-85. [Crossref] [PubMed]
- Takada K, Takamori S, Brunetti L, et al. Impact of Neoadjuvant Immune Checkpoint Inhibitors on Surgery and Perioperative Complications in Patients With Non-small-cell Lung Cancer: A Systematic Review. Clin Lung Cancer 2023;24:581-590.e5. [Crossref] [PubMed]
- Henschke CI, Yip R, Sun Q, et al. Prospective Cohort Study to Compare Long-Term Lung Cancer-Specific and All-Cause Survival of Clinical Early Stage (T1a-b; ≤20 mm) NSCLC Treated by Stereotactic Body Radiation Therapy and Surgery. J Thorac Oncol 2024;19:476-90. [Crossref] [PubMed]
- Shi Y, Ma X, He D, et al. Neoadjuvant SBRT combined with immunotherapy in NSCLC: from mechanisms to therapy. Front Immunol 2023;14:1213222. [Crossref] [PubMed]
- Lee JM, Kim AW, Marjanski T, et al. Important Surgical and Clinical End Points in Neoadjuvant Immunotherapy Trials in Resectable NSCLC. JTO Clin Res Rep 2021;2:100221. [Crossref] [PubMed]